Introduction
Morchella esculenta (Morel) is one of the wild species that is significant and economically valuable hence recognized as “growing gold of mountains”.1 It is one of the most extensive and profitable international commerce networks now globally significant.2 It is believed that in 2023, the market size of the India mushroom was USD 1.18 billion and from 2024 to 2030, the compound annual growth rate (CAGR) is expected to grow at 12.7% from 2024 to 2030. The rising demand for a protein-rich diet among the growing vegan population is anticipated to have a major impact on the market. Mushroom is referred to as a superfood due to its high nutrient content. Selenium, vitamin D, glutathione, and ergothioneine are important nutrients found in large quantity in mushrooms. These nutrients help prevent or lessen chronic diseases like dementia, cancer, and heart disease by lowering oxidative stress.3 In India, the demand for spawns is about 8000-10000 tons per annum based on the production statistics.4 With an annual yield of 487 thousand tonnes, or 2% of the global total, India was one of the leading producers of mushrooms in the world as of 2020.3 More than 300 types of wild mushrooms can be found in India. The FAO Statistics-2022 ranking of the world’s mushroom output (in tonnes) was evaluated i.e. China (5,150,000)> Italy (785,000)> USA (388,450) > Netherlands (307,000)> Poland (220,000) and India (29992).5 However, India consumes fewer than 50 grams of mushrooms per person, compared to nearly a kilogramme in other nations.
This usually occurs in thick coniferous forests (2500–3500 msl) with loamy soil that is rich in humus from March to July.1,6 It is commonly found in a saprobic or mycorrhizal relationship with coniferous trees and hardwood.7 Morels can be found in a variety of habitats and conditions, including cold soils, dead trees, and disturbed soil. Additionally, they have adapted to survive in a range of environments, including dunes, river bottoms, and abandoned mines.8-10 Although they are uncommon in warmer temperatures, morels can be found in all temperate and boreal forests in the Northern Hemisphere as well as some Mediterranean and subtropical areas.11 The woodlands of Jammu and Kashmir and Himachal Pradesh in India are home to this mushroom, which is indigenous to the Kullu District.
The scientific classification of this mushroom is: Fungi>Ascomycota>Pezizomycetes>Pezizales> Morchellaceae>Morchella>esculenta/ conica/ deliciosa/ angusticeps/ arassipes/ hybrida=semilibera. Morchella esculenta, M. conica, M. deliciosa, M. angusticeps, M. arassipes and M. hybrid (M. semilibera) are reported from India.12 Because of its distinctive flavour and high nutritional content, M. esculenta is a pricey commodity.13 Fruit bodies start as grayish sponges, expand to yellowish sponges with pits and ridges on white stems: pitted caps, 2-10 cm tall, 7 cm broad, fuse to stem. The crown can be yellow to tan, pale brown to greyish brown, or pale brownish cream.14,15
This versatile ingredient, rich in vitamins, minerals, proteins, carbohydrates, fibers, and aromatic compounds, is used in various recipes worldwide due to its unique flavor, and texture, and antioxidant, anticancer, antibacterial, and anti-inflammatory are among its many pharmacological characteristics. When used improperly, it can cause numerous negative effects and be toxic if consumed uncooked. This mushroom’s extracts show promise as treatments for diabetes, heart disease, and cancer16 and colon cancer, one of the cancers that kill people.17
Nutritive value
The edible fruiting body of M. esculenta contains many active compounds, including organic acids, carotenoids, tocopherols, and phenolic compounds. In the carotenoids, lycopene and β-carotene are present. The tocopherols include δ-, α-, and γ-tocopherol, while the phenolic compounds comprise protocatechuic acid, p-coumaric acid, and p-hydroxybenzoic acid. Additionally, organic acids such as citric, oxalic, fumaric, quinic, and malic acid are found in this mushroom.
This mushroom is both tasty and nutritious. It is rich in protein, carbohydrates, and vitamins—especially those in the B-complex—with smaller amounts of vitamins A, C, and D. It also supplies essential minerals like calcium, iron, copper, zinc, magnesium, manganese, sodium, phosphorus, potassium, and selenium. With few calories and low fat,18,19 its composition is approximately 38% carbohydrates, 32.7% protein, 17.6% fiber, 9.7% ash, and 2.0% fat. Moreover, it has been reported to contain around 195 mg/g of iron, 98.9 mg/g of zinc, 62.6 mg/g of copper, 54.7 mg/g of manganese, 23.5 mg/g of potassium, 3.49 mg/g of phosphorus, 1.82 mg/g of magnesium, 0.85 mg/g of calcium, and 0.18 mg/g of sodium.20,21
Previous studies have also detected various aromatic compounds such as phenols, alcohols, acids, esters, aldehydes, ketones, and terpenes in M. esculenta. The main aromatic substances identified include alcohol (15.55%), carbamic acid (11.37%), phenol (50.88%), and esters.22 Additionally, proteins derived from the mycelium of M. esculenta can serve as a useful protein supplement, similar to the proteins found in the fruiting body.23 Due to its high protein content, this mushroom is easier to digest compared to many other vegetables.
Furthermore, M. esculenta is believed to help treat conditions such as hepatitis B, colds, stomach aches, and headaches. It may also lower cholesterol levels, ease sleep problems and fatigue, regulate blood sugar, and provide an effective remedy for anemia.24-31
Historical Developments
The first documented instance of outdoor cultivation dates back to 1882 in France, where M. esculenta was cultivated alongside Jerusalem artichokes.32 Subsequent advancements were achieved through patents issued between 1986 and 1989, which optimized sclerotia-based inoculation and regulated environmental parameters such as humidity and temperature.33,34 Despite initial success, attempts at indoor cultivation were abandoned in 2008 due to contamination and yield inconsistencies.35 Innovations such as ectomycorrhizal symbiosis36 and controlled soilless systems37 have since been introduced. Notably, large-scale field cultivation in China has seen significant expansion, with yields reaching 7620 Kg/ha and over 1600 hectares cultivated by 2016.38 The artificial cultivation of morels has grown rapidly, with China leading advancements in field soil culture since 2012, achieving significant scale and yield. However, challenges like yield instability, genetic variability, and contamination have driven interest in cultivating morel mycelia as an alternative.39
Life Cycle
Morel mushrooms, members of the ascomycetes, reproduce through both sexual and asexual means. In the sexual phase, the fruiting body or ascocarp, houses numerous sac-like structures known as asci, each usually containing eight spores. A thorough understanding of these reproductive processes is essential for the successful artificial cultivation of morels.40 Key stages in their life cycle include forming sclerotia and producing conidia. (Fig.-1) illustrates the asexual reproduction of Morchella esculenta via conidia, drawing parallels with powdery mildew and emphasizing recombination and propagation mechanisms that enhance resilience under stressful conditions. Additionally, the ascospores that are released give rise to the primary mycelium, which can subsequently develop sclerotia to survive under adverse conditions.40 In spring, sclerotia can germinate in two ways: myceliogenically, leading to the formation of a new primary mycelium, or carpogenically, resulting in the development of a fruiting body. Moreover, when one primary mycelium meets a compatible counterpart, their hyphae merge to form a heterokaryon containing paired nuclei. In morels, the sclerotia are actually pseudosclerotia, formed through the repeated branching and expansion of terminal hyphae. These hyphae may be either primary (homokaryotic) or secondary (heterokaryotic).41 Conidia in Morchella esculenta function as asexual spores, aiding both mating and colony expansion. Acting as gametes and propagules, they are influenced by environmental factors like air, nutrients, and moisture. Their formation is crucial for the its reproduction and cultivation.42
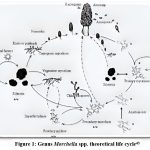 |
Figure 1: Genus Morchella spp. theoretical life cycle43 |
The growth substrates along with the nutritional composition affects the both mycelial characteristics and sclerotia formation.44,45 While the trophic strategies of Morchella vary, available evidence suggests that the genus comprises facultative mycorrhizal, saprophytic, as well as strictly mycorrhizal species. However, these findings do not indicate that an ectomycorrhizal association is essential for the morel life cycle or for ascocarp production.46
In China, growers assess the quality of morel spawns by evaluating the amount of sclerotia produced. Due to the rapid senescence of morel strains, which results in a loss of vigor and viability, it becomes necessary to continually reselect strains from spore cultures.46 Successful outdoor cultivation requires an exogenous nutrient supply, although the mechanism by which these nutrients affect growth remains unclear. A transition from a nutrient-rich to a nutrient-poor environment prompts the fungus to shift into its sexual growth phase, during which ascocarps are formed.47
Most morel cultivators in China are of the opinion that exogenous nutrient bags provide the essential nourishment required for mycelial growth.48 Providing exogenous nutrients involves considerable labor and expense, including the preparation, sterilization, and incorporation of the nutrient into the mushroom bed. Consequently, focusing on the mechanization of this nutrient supply—or exploring alternative cultivation methods based on its underlying mechanisms—represents a promising direction for improvement.39,48
Properties and the Applications of Morchella esculenta
Overview and Toxicity
Morchella esculenta is valued for its culinary and medicinal uses, benefiting conditions like cardiovascular diseases, diabetes, cancer, and obesity. However, improper preparation can cause neurotoxic effects, such as ataxia and visual disturbances, which are neutralized by adequate cooking.12,49
Medicinal and Nutritional Benefits
Rich in polysaccharides, proteins, vitamins, and trace elements, M. esculenta provides significant health benefits. Its bioactive compounds, including phenolics, offer antioxidant, anti-inflammation and antitumor effects.50,51 M. esculenta is rich in the essential amino acids, wherein glutamic acid being the most predominant.27
Antioxidant Properties
M. esculenta has potent antioxidant activity through phenolic compounds, tocopherols, and organic acids, which reduce oxidative stress.52 Serbian samples show strong DPPH radical scavenging and polysaccharides from fermentation scavenge superoxide and hydroxyl radicals.53 (MEPs) M. esculenta polysaccharides enhance antioxidant enzyme activities, reduce lipid peroxidation by decreasing (MDA) Malondialdehyde levels, and also activate the AKT/Nrf2 signalling pathway, enhancing protective proteins like HO-1 to help protect against oxidative damage.54,55 They also mitigate nephrotoxicity and liver damage induced by oxidative stress.56
Antimicrobial Potential
M. esculenta exhibits a lot of antimicrobial activity against bacteria, especially those which are already resistant to antibiotics such as the Salmonella and Staphylococcus aureus. Badshah et al. and Kalyoncu et al. affirmed sesquiterpenes and steroids among the active principles hence corroborating its traditional use to infection control are proven effector compounds.57,58 However, it is not the only evidence that the plant is effective in controlling these infections microbiologically. Additionally, M. esculenta has been found to inhibit the growth of E. coli and exhibits effectiveness against both Gram-positive bacteria and also the Gram-negative bacteria which is supported by scientific evidence such as that mentioned by.59,60
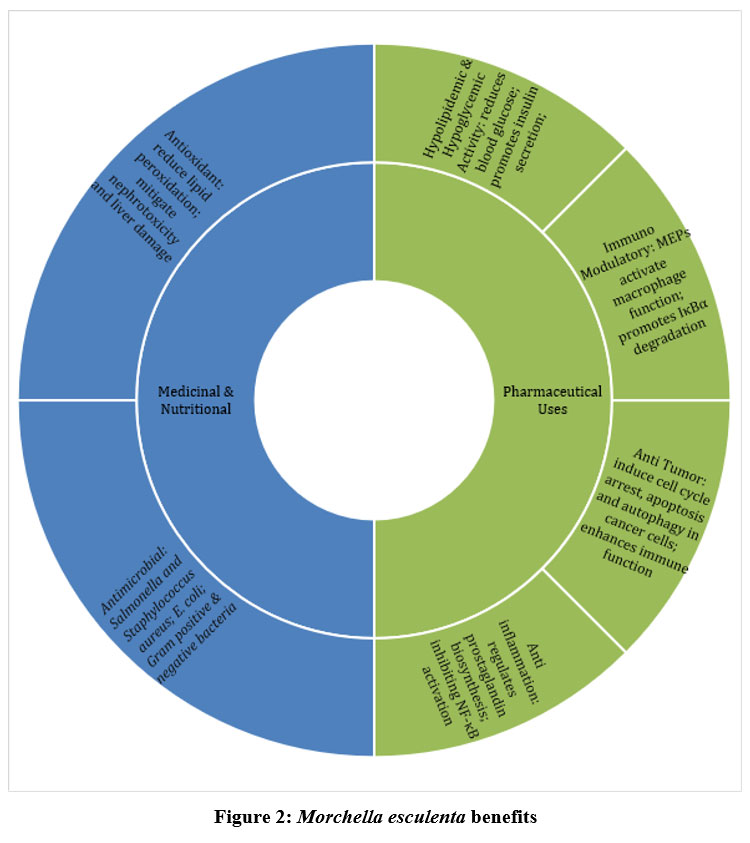 |
Figure 2: Morchella esculenta benefits |
Uses as Pharmaceuticals
Morchella species contain diverse bioactive and nutritional compounds and are valued by traditional hilly societies for their healthcare applications.13,52,61-63 M. esculenta has been traditionally employed to alleviate excessive phlegm, aid digestion, and support heart health,64 stomach aches, wound healing,65 and also function as a emollient and laxative.31 Additionally, it alleviates type 2 diabetes in mice by improving gut function, glucose tolerance, and reducing inflammation.66
Anti-inflammation Effects
The methanol extract derived from the entire plant exhibits anti-inflammatory properties and aids in pain reduction. It inhibits both acute and chronic inflammation.26,28 Sulfated MEPs protect against inflammation by inhibiting NF-κB activation.67 The active compound, (EOYA) (E)-Octadec-11-En-9-Ynoic Acid, interacts with proteins such as NR1I2, PTGS1, and PTGS2, regulating prostaglandin biosynthesis through the arachidonic acid pathway.68
Antitumor Activity
Antitumor activities are exhibited by polysaccharides derived from M. esculenta. These polysaccharides induce cell cycle arrest, apoptosis and autophagy in cancer cells, including the HT-29 human colon cancer cells.69,70 In M. esculenta, the mycelia contain chemicals that have the potential to regulate the progression of cancer at several stages or to operate at the same stage concurrently. The extracted polysaccharide from the M. esculenta fruiting bodies has shown immunological stimulatory action.20 Furthermore, MEPs enhance immune function, stimulate macrophages, and mitigate chemotherapy side effects.71
Immuno-modulatory Activities
Active immunostimulatory properties are exhibited by the galactomannan polysaccharide that was isolated from M. esculenta.24 MEPs activate macrophage function by binding to TLR4 receptors and stimulating signalling pathways like NF-κB, JNK, and Akt, which boosts phagocytosis and cytokine production.67 Acetylated MEP modulates macrophage immune activation by promoting IκBα degradation and activating NF-κB and MAPK pathways, enhancing viability and nitric oxide production.55
Hypolipidemic and Hypoglycemic Activity
Several MEPs show strong hypolipidemic and hypoglycemic effects. An acidic polysaccharide from M. esculenta reduces blood glucose and promotes insulin secretion in diabetic rats. MEPs also exhibit anti-atherosclerotic effects by lowering serum total and LDL cholesterol, and triglycerides via the AMPKα signaling pathway.72 Carboxymethyl FB-MEP enhances cholesterol-lowering by upregulating CYP7A1 and LDL-R expression and downregulating HMG-CoA in the liver. Both FB-MEP treatment and carboxymethylated FB-MEP reduce serum TC and TG, while increasing HDL-C, indicating significant lipid-lowering activity.73 The benefits of Morchella esculenta have been depicted in brief in the Fig.-2.
Cultivation of Morchella esculenta
Morchella esculenta is a saprotrophic fungus that predominantly colonizes humus-rich forest floors, where it forms dense mycelial networks under adequate nutrient availability. Ascocarps emerge post-precipitation, favoring ecosystems dominated by tree species such as Cedrus deodara and Pinus wallichiana, alongside medicinal flora including Angelica glauca and Dactylorhiza hatagirea. Harvesting occurs during the peak season from April to June.13,74 Due to the impracticality of traditional cultivation methods, submerged fermentation has been explored as an alternative.75
Cultivation Techniques
Three main systems include Ower’s indoor cultivation (focused on sclerotia production and fruiting induction), Yunnan’s stump wood bionic cultivation (utilizing wood nutrients but limited by forest resource use), and field cultivation in China (using nutrient bags to enhance stability and yield).76
Spawn Production
The production of high-quality spawn is fundamental to successful cultivation. Cultures are typically initiated from healthy fruit bodies or commercially obtained isolates and propagated on (PDA) potato dextrose agar supplemented with humus. Common substrates include wheat, wheat bran, sawdust, and gypsum, with a recommended composition of wheat (46%), husk (20%), and humus (4%).77 Approximately 4500 spawn bags (14–28 cm) are required per hectare, incurring costs between $7620 and $10,880.
Spawning
Optimal spawning conditions necessitate loose, aerated soil with a maintained humidity of 50–70%. Spawning typically occurs between October and December when local temperatures are below 20°C. Spawn is sown in 15 cm-deep beds, covered with a 3–5 cm layer of casing soil. Protective measures such as mulching and canopy installation ensure stability in temperature and moisture levels.78
Exogenous Nutrition
The application of exogenous nutrition bags, containing substrates like wheat and sawdust, facilitates robust mycelial development. These bags are placed at 50 cm intervals and deplete within 40–45 days, effectively supporting ascocarp formation.79,80
Fruiting Management
Critical environmental parameters include soil moisture (>50%) and air humidity (85–90%). Flooding practices have been shown to stimulate primordium differentiation by promoting interactions with beneficial soil microbiota. Optimal fruiting temperatures range from 6–10°C, with diurnal variations enhancing primordium initiation. Pest management relies on biological control methods to mitigate competitive fungi and insect infestations.76,81
Artificial Cultivation
Morchella species were artificially cultivated in 2012, began in Sichuan of China. It has since grown to yields of up to 15,000 kg per hectare. It’s success relies on good quality spawn and selection of suitable species and also prevention from uncontrollable species like M. angusticeps. Despite continuous cropping leading to variation in the properties of soils and crop diseases in some regions, challenges for food security such as erratic yields and reduced production are still prevalent. Therefore, the crop rotation, soil alterations and disease resistant strains are being investigated for improved sustainability.38 Although morel cultivation is profitable, environmental variability and limited knowledge about the most suitable conditions for growth still hinder its development.76,82
Harvesting and Market Significance
Ascocarps, measuring 10–15 cm, are harvested upon maturity and subsequently dried for commercial distribution. In India, M. esculenta fetches premium prices, ranging from ₹14,000 to ₹15,000/Kg, largely driven by its short harvesting window and high demand. However, overharvesting has led to significant population declines in natural habitats, emphasizing the need for sustainable harvesting practices.30,83
Sustainable Farming
To address soil health and production concerns, sustainable morel farming in continuous cropping settings emphasizes disinfection of the soil, rotations of the crop, intercropping, deep ploughing of the soil, breeding, and microbial inoculants. Soil disinfection methods like solarization and fumigation are also effective.84
Genesis of Morel mushrooms continuous cropping obstacles
Soil’s Physicochemical Properties Modifications
Morchella spp. is a saprotrophic fungus that effectively breakdown complicated molecules into simpler ones by secreting various enzymes.85 This action leads to a rise in nutrient levels in the soil.85,86 Thus, continuous morel cropping causes the soil to accumulate nutrients, which makes it unsuitable for fruiting and lowers the yields of primordium and fruiting bodies.87
Soil microbial population alterations
Morchella operates as a highly invasive species that quickly controls the soil. The competition for nutrients and space with the native soil microbial communities leads to the alteration of the native resident fungal communities.84
Soil-borne Disease
High amounts of fungal infections of morel mushrooms are seen in the soil bed during the fruiting body stage of continuous cropping.88 Consequently, the production of morel mushrooms decreases, and soil-borne diseases become more prevalent.89 Potential morel mushroom pathogens that were found in high concentrations include Gibberella, Microidium, Trichoderma, Penicillium, Sarocladium and Streptomyces. In addition to these, the Aspergillus and Fusarium were found in low concentrations.87,90
Allelopathy and Autotoxicity
The phenolic acid extracts from the soils show allelopathic effects, which promote the growth of harmful fungus while simultaneously suppressing the growth of morel mushrooms.89 The production of endogenous organic acids by Ganoderma lucidum results in stress, which in turn triggers autotoxicity feedback and cropping hurdles.91
Strategies for Growing Morels Sustainably in Continuous Cropping Environments
Deep Ploughing
Deep ploughing helps in decreasing physicochemical properties by nutrient-deficient deeper layers of soil to the surface. It also helps in the restoration of microbial diversity in the soil.89
Crop Rotation and intercropping
The yield and quality of the morel mushroom’s fruit body were enhanced when it was rotated with rice.92 Water has a high heat conductivity, irrigating the soil with enough water raises its temperature. Consequently, this aids in clearing the soil of autotoxic and allelopathic compounds.93-95 By improving the annual yield per unit of land area and facilitating more effective land use, intercropping eventually boosts economic benefits.94
Soil Disinfection
Physical disinfection techniques include radiation sterilization, steam disinfection, microwave therapy, and solarisation.96 Greenhouse Thermal pasteurization, a type of solarisation that raises the temperature and aids in the removal of harmful microbes, is used by farmers in China.97
Breeding of Morel Mushrooms
By using monokaryotic hybridization, new Morel mushroom isolates are produced which improves the variety by increasing yield and quality.97,98
Microbial Inoculants
Many microbes inoculants are used as biofertilizers to perform specific functions, as biopesticides for the management of various diseases, and in bioremediation of various contaminants.99,100
Competitive Landscape and Mushroom Cultivation Market Share Analysis
The mushroom cultivation industry is characterized by a competitive landscape that provides insights into various market players. Key aspects include company profiles, financial performance, revenue generation, market potential, research and development investments, expansion strategies, global presence, production facilities, manufacturing capacities, strengths and weaknesses, product launches, product range, and market dominance in different applications. The information presented focuses specifically on companies engaged in the mushroom farming sector.3,30,83
Several key participants involved in the mushroom farming industry include:
| 1. Monaghan Group (Ireland) | 2. Costa (India), Bonduelle (France) |
| 3. Walsh Mushrooms Group (Ireland) | 4. Monterey Mushrooms (U.S.) |
| 5. Mycelia (Belgium) | 6. Cargill, Incorporated (U.S.) |
| 7. Smithy Mushrooms Ltd. (U.K.) | 8. Myco Terra Farm (Belgium) |
| 9. Rheinische Pilz Zentrale GmbH (Germany) | 10. Lambert Spawn (U.S.) |
| 11. Italspwan (Italy) | 12. The Greenyard (Belgium) |
| 13. Mushroom SAS (Italy) | 14. Heereco (Netherlands) |
| 15. Hirano Mushroom LLC (Kosovo) | 16. Bluff City Fungi (U.S.) |
| 17. Fujishukin Co. Ltd. (Japan) | 18. Smithy Mushrooms (U.K.) |
Conclusion
Due to its high market value, the morel mushroom plays a crucial role in a country’s economy. Morel mushrooms, belonging to the genus Morchella, are globally distributed and hold significant social and economic importance. These mushrooms are highly valued as edible fungi, with substantial harvests occurring when certain species fruit abundantly, particularly in the first year following a fire. However, despite their widespread use, limited research has quantified post-fire morel mushroom abundance. M. esculenta contains functional polysaccharides beneficial to human health. Scientific advancements are essential for improving its cultivation and ensuring the availability of viable propagules. A collaborative approach involving mycologists is necessary to identify suitable microcultures for field applications, while agronomists and ecologists must investigate resource management mechanisms. Comprehensive research is required in systematic, ecological, physiological, and genetic aspects of morel mushrooms. Detailed studies on their lifecycle, including spore formation, dispersal, and germination, are essential. Additionally, analysis of the soil’s physicochemical properties beneath the fruiting bodies is crucial to understanding the optimal conditions for spore germination. Determining the ideal factors for sclerotia formation, growth, and maturation, as well as their regulation, is vital. Furthermore, proper identification and documentation of plant species associated with Morchella fruiting bodies will enhance understanding of their ecological interactions.
Acknowledgment
I want to express my sincere gratitude to all those who have contributed to completing this review article. Special thanks to Banaras Hindu University for providing me with an excellent work environment. Lastly, I would like to thank my family and friends for their constant support and encouragement during this endeavor.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Author Contributions
Saroj Belbase and Jiwan Paudel: the conceptualization, literature search and wrote the original draft.
Shradha Subba: analysis, review, and editing and revised the manuscript.
Surush Das: review and editing of the final manuscript
Shrvan Kumar: the critical insights and supervision of the overall writing
Reference
- Ali, H., Sannai, J., Sher, H. and Rashid. Ethnobotanical profile of some plant resources in Malam Jabba valley of Swat, Pakistan. J. Med. Plants Res. 2011; 5(17), 4171-4180.
- Thompson V.V. Morel: a lifetime pursuit, Glencoe, MO: Missouri Mycological Society; 1994.
- India Mushroom Market Size, Share & Trends Analysis Report By Product (Button, Shiitake, Oyster), By Form, By Distribution Channel, By Application (Food, Pharmaceuticals, Cosmetics), And Segment Forecasts, 2024 – 2030, Grand View Report. 2024; Report ID: GVR-4-68040-209-4. pp.1-75.
- Sharma, V. P., Annepu, S. K., Gautam, Y., Singh, M., & Kamal, S. Status of mushroom production in India. Mushroom Research, 2017; 26(2), 111-120.
- http://www.fao.org/faostat/en/#data. 2002; Retrieved 2024-01-18.
- Wagay, J.A. and D. Vyas. Phenolic quantification and antioxidant activity of Morchella esculenta. J. Pharma Bio. Sci.. 2011; 2(1): 188 – 197.
- Hamayun, M., Khan, S. A., Ahmad, H., Shin, D. H., & Lee, I. J. Morel collection and marketing: A case study from the Hindu-Kush mountain region of Swat, Pakistan. Lyonia. 2006;11, 7-13.
- Kaul T.N. Studies of the genus Morchella in Jammu and Kashmir I. Soil composition in relation to carpophore development. Bulletin of the Botantical Society of Bengal. 1975;29: 127–134.
- Kuo M. When and where morels grow. 2002; http://www. mushroomexpert.com/morels/whenwhere.html.;
- Weber N.S. A morel hunter’s companion: a guide to the true and false morels of Michigan. MI: TwoPeninsula Press, Lansing. 1988; URL- http://agris.fao.org/agris-search/search.do?recordID=US9153184
- Wedin M, Döring H, Gilenstam G. Saprotrophy and lichenization as options for the same fungal species on different substrata: environmental plasticity and fungal lifestyles in the Stictis–Conotrema complex. New Phytologist. 2004;164(3):459-465. doi:10.1111/j.1469-8137.2004.01198.x
CrossRef - Nautiyal S, Maikhuri RK, Rao KS, Saxena KG. Medicinal plant resources in Nanda Devi Biosphere Reserve in the Central Himalayas. Journal of Herbs Spices & Medicinal Plants. 2001;8(4):47-64. doi:10.1300/j044v08n04_06
CrossRef - Wasser SP, Weis AL. Medicinal Properties of substances occurring in higher basidiomycetes mushrooms: Current Perspectives (Review). International Journal of Medicinal Mushrooms. 1999;1(1):31-62. doi:10.1615/intjmedmushrooms.v1.i1.30
CrossRef - Ammirati J.F., McKenny M., Stuntz D.E. The New Savory Wild Mushroom. Seattle: University of Washington Press. 1987;pp. 209–10. ISBN0-295-96480-4.
- Roody WC. Mushrooms of West Virginia and the Central Appalachians. Lexington, Kentucky: University Press of Kentucky. 2003; p. 485. ISBN 0-8131-9039-8.
- Guillamon E, García-Lafuente A, Lozano M, et al. Edible mushrooms: Role in the prevention of cardiovascular diseases. Fitoterapia. 2010;81(7):715-723. doi:10.1016/j.fitote.2010.06.005
CrossRef - Saito H, Miyata E, Sasaki T. A 13C Nuclear magnetic resonance study of Gel-Forming (1→3)-Β-D-Glucans: Molecular-Weight dependence of helical conformation and of the presence of junction zones for association of primary molecules. Macromolecules. 1978;11(6):1244-1251. doi:10.1021/ma60066a035
CrossRef - Mattila P, Könkö K, Eurola M, et al. Contents of vitamins, mineral elements, and some phenolic compounds in cultivated mushrooms. Journal of Agricultural and Food Chemistry. 2001;49(5):2343-2348. doi:10.1021/jf001525d
CrossRef - Negi, C.S. Morels (Morchella) in Kumaun Himalaya. Nat. prod. Rad.. 2006;5(4): 306 – 310.
- Raman V.K., Saini M., Sharma A and Parashar B. Morchella esculenta: A herbal boon to pharmacology. International Journal of Development Research. 2018; 08(3): 19660-65.
- Wahid, M., Sattar, A. and Khan, S. (1988). Composition of wild and cultivated mushrooms of Pakistan. Mushroom J Trop.. 1988; 8(2): 47 – 51.
- Genccelep H, Uzun Y, Tunçtürk Y, Demirel K. Determination of mineral contents of wild-grown edible mushrooms. Food Chemistry. 2009;113(4):1033-1036. doi:10.1016/j.foodchem.2008.08.058
CrossRef - Taskin H. Detection of volatile aroma compounds of morchella by Headspace Gas Chromatography Mass Spectrometry (HS-GC/MS). Notulae Botanicae Horti Agrobotanici Cluj-Napoca. 2013;41(1):122. doi:10.15835/nbha4118344
CrossRef - Duncan CJG, Pugh N, Pasco DS, Ross SA. Isolation of a Galactomannan That Enhances Macrophage Activation from the Edible Fungus Morchella esculenta. Journal of Agricultural and Food Chemistry. 2002;50(20):5683-5685. doi:10.1021/jf020267c
CrossRef - Janardhanan, K. K., Kaul, T. N. and Husain, A. Use of vegetable waste for the production of fungal protein from Morchella J. Food Sci. Technol.. 1970;7: 197–199.
- Kumar S, Ziereis K, Wiegrebe W, Müller K. Medicinal plants from Nepal: evaluation as inhibitors of leukotriene biosynthesis. Journal of Ethnopharmacology. 2000;70(3):191-195. doi:10.1016/s0378-8741(00)00203-8
CrossRef - Li IC, Chiang LH, Wu SY, Shih YC, Chen CC. Nutrition Profile and Animal-Tested Safety of Morchella esculenta Mycelia Produced by Fermentation in Bioreactors. Foods. 2022;11(10):1385. doi:10.3390/foods11101385
CrossRef - Nitha, B., Meera, C.R. and Janardhanan, K.K. Antiinflammatory and antitumour activities of cultured mycelium of morel mushroom, Morchella esculenta. Curr 2007;92: 235–239.
- Sharma, A., Sharma, S., Chandel, S., Vatsa, E. and Dr. Parashar, B. A review on Morchella esculanta: therapeutically potent plant., World J. of Pharm. and Pharmac. Sci. 2016; 5(9): 685-699.
- Sharma, B.M., Singh, B.M. and Pal, J. Ascocarp development of Morchella esculenta under natural conditions. In: Proceedings National Symposium, “Recent Trends in the Management of Biotic and Abiotic stress in Plants“, HPKV, Palampur, Nov., 2-3, 1995.
- Sher, H., Elyemeni, M., Sher, H., & Hussain, K. Ethnobotanical and economic observations of some plant resources from the northern parts of Pakistan. Ethnobotany research and applications. 2011; 9, 027-041.
CrossRef - Roze, M. E. Adherence de la base d’appareils ascospores de Morchella sur Helianthus tuberosus. Bull Soc Bot Fr. 1882; 19, 166-167.
- Ower RD, Mills GL, Malachowski JA, inventors; Neogen Corporation, assignee et al. Cultivation of Morchella. United States patent US 4,594,809. 17 Jun, 1986.
- Pilz D., Rebecca M.L., Susan A., et al. Ecology and management of morels harvested from the forests of western North America. Portland (OR): US Department of Agriculture, Forest Service, Pacific Northwest Research Station; 2007.
CrossRef - Tan F.H. History, current station and prospect of Morel cultivation. Edible Med Mushrooms (Chinese). 2016; 24:140–144.
- Miller S.C., inventor; Miller S.C., assignee. Cultivation of Morchella. United States patent US 6,907,691B2. 21 Jun, 2005.
- Masaphy S. Biotechnology of morel mushrooms: successful fruiting body formation and development in a soilless system. Biotechnology Letters. 2010;32(10):1523-1527. doi:10.1007/s10529-010-0328-3
CrossRef - Liu Q, Ma H, Zhang Y, Dong C. Artificial cultivation of true morels: current state, issues and perspectives. Critical Reviews in Biotechnology. 2018;38(2):259-271. doi:10.1080/07388551.2017.1333082
CrossRef - Li QM, Zhang D, Zhang JZ, et al. Crop rotations increased soil ecosystem multifunctionality by improving keystone taxa and soil properties in potatoes. Frontiers in Microbiology. 2023;14. doi:10.3389/fmicb.2023.1034761
CrossRef - Volk TJ, Leonard TJ. Cytology of the life-cycle of Morchella. Mycological Research. 1990;94(3):399-406. doi:10.1016/s0953-7562(09)80365-1
CrossRef - Volk TJ, Leonard TJ. Experimental studies on the Morel. I. heterokaryon formation between monoascosporous strains of Morchella. Mycologia. 1989;81(4):523-531. doi:10.1080/00275514.1989.12025783
CrossRef - Liu W, He P, Zhang J, et al. Ultrastructure and physiological characterization of Morchella mitospores and their relevance in the understanding of the Morel life cycle. Microorganisms. 2023;11(2):345. doi:10.3390/microorganisms11020345
CrossRef - Alvarado-Castillo, G., Mata, G., & Sangabriel-Conde, W. Entendiendo el ciclo de vida de las morillas (Morchella). Revista mexicana de micología. 2014;40, 47-50.
- Stott, K., & Mohammed, C. Specialty mushroom production systems: maitake and morels. Australia: RIRDC. 2004.
- Winder RS. Cultural studies of Morchella Mycological Research. 2006;110(5):612-623. doi:10.1016/j.mycres.2006.02.003
CrossRef - Stamets P. Growing gourmet and medicinal mushrooms. Berkeley (CA): Ten Speed Press. 2000; p. 574.
- Ower R.D., deceased, Mills G.L., Malachowski J.A., inventors; Neogen Corporation, assignee. Cultivation of Morchella. United States patent US 4,866,878. 19 Sep, 1989.
- Zhao YC, Cai HM, Zhang XL. Difficulties and prospect of Morel commercialization in China. Edible Med Mushrooms (Chinese). 2016;24:133–139.
- Fayaz, A.L., Lone, S., Aziz, M.A. and Malla, F.A. Ethnobotanical Studies in the Tribal areas of district Kupwara, Kashmir, India. J. Pharma Bio Sci. 2012;3(4): 399 – 411.
- Elmastas M, Turkekul I, Ozturk L, Gulcin I, Isildak O, Aboul-Enein H. Antioxidant Activity of Two Wild Edible Mushrooms (Morchella vulgaris and Morchella esculanta) from North Turkey. Combinatorial Chemistry & High Throughput Screening. 2006;9(6):443-448. doi:10.2174/138620706777698544
CrossRef - Nitha B, Fijesh PV, Janardhanan KK. Hepatoprotective activity of cultured mycelium of Morel mushroom, Morchella esculenta. Experimental and Toxicologic Pathology. 2013;65(1-2):105-112. doi:10.1016/j.etp.2011.06.007
CrossRef - Ferreira I, Barros L, Abreu R. Antioxidants in wild mushrooms. Current Medicinal Chemistry. 2009;16(12):1543-1560. doi:10.2174/092986709787909587
CrossRef - Li X, Sun Q, Li S, et al. Production with Fermentation Culture and Antioxidant Activity of Polysaccharides from Morchella esculenta. Fermentation. 2024;10(1):46. doi:10.3390/fermentation10010046
CrossRef - Chen S, Wang M, Veeraperumal S, et al. Antioxidative and Protective Effect of Morchella esculenta against Dextran Sulfate Sodium-Induced Alterations in Liver. Foods. 2023;12(5):1115. doi:10.3390/foods12051115
CrossRef - Wu H, Chen J, Li J, Liu Y, Park HJ, Yang L. Recent Advances on Bioactive Ingredients of Morchella esculenta. Applied Biochemistry and Biotechnology. 2021;193(12):4197-4213. doi:10.1007/s12010-021-03670-1
CrossRef - Nitha B, Janardhanan KK. Aqueous-ethanolic extract of morel mushroom mycelium Morchella esculenta, protects cisplatin and gentamicin induced nephrotoxicity in mice. Food and Chemical Toxicology. 2008;46(9):3193-3199. doi:10.1016/j.fct.2008.07.007
CrossRef - Badshah H, Qureshi RA, Khan J, et al. Pharmacological screening of Morchella esculenta (L.) Pers., Calvatia gigantea (Batsch ex Pers.) Lloyd and Astraeus hygrometricus Pers., mushroom collected from South Waziristan (FATA.). Journal of Medicinal Plants Research. 2012;6(10):1853-1859. doi:10.5897/jmpr11.1138
- Kalyoncu F, Oskay M, Sağlam H, Erdoğan TF, Tamer AÜ. Antimicrobial and antioxidant activities of mycelia of 10 wild mushroom species. Journal of Medicinal Food. 2010;13(2):415-419. doi:10.1089/jmf.2009.0090
CrossRef - Canli K, Benek A, Şenturan M, Akata İ, Altuner EM. In vitro Antimicrobial Activity of Morchella esculenta and Trametes versicolor. Mantar Dergisi. 2019;10(3):28-33. doi:10.30708/mantar.622863
- Shameem N, Kamili AN, Ahmad M, Masoodi FA, Parray JA. Antimicrobial activity of crude fractions and morel compounds from wild edible mushrooms of North western Himalaya. Microbial Pathogenesis. 2017;105:356-360. doi:10.1016/j.micpath.2017.03.005
CrossRef - Alves M, Ferreira I, Dias J, Teixeira V, Martins A, Pintado M. A review on antimicrobial activity of mushroom (Basidiomycetes) extracts and isolated compounds. Planta Medica. 2012;78(16):1707-1718. doi:10.1055/s-0032-1315370
CrossRef - Ferreira, I.C.F.R., Vaz, J.A., Vasconcelos, M.H. and. Martins, A. Compounds from wild mushrooms with antitumour potential. Anticancer Agents Med. Chem.. 2010;10(5): 424 – 436.
CrossRef - Prasad, P., Chauhan, K., Kandari, L. S., Maikhuri, R. K., Purohit, A., Bhatt, R. P., & Rao, K. S. Morchella esculenta (Guchhi): Need for scientific intervention for its cultivation in Central Himalaya. Current Science. 2002; 82(9), 1098-1100.
- Ying, J., Mao, X., Ma, Q., Zong, Y. and Wen, H. Icones of Medicinal Fungi from China, Xu, Y., Trans., Science Press: Beijing 1987;pp.38 – 45.
- Mahmood, A., Malik, R.N., Shinwari, Z.K. and Mahmood, A. Ethnobotanical survey of plants from Neelum, Azad jammu &kashmir, Pak. J. Bot. 2011;43: 105 – 110.
- Rehman AU, Siddiqui NZ, Farooqui NA, et al. Morchella esculenta mushroom polysaccharide attenuates diabetes and modulates intestinal permeability and gut microbiota in a type 2 diabetic mice model. Frontiers in Nutrition. 2022;9. doi:10.3389/fnut.2022.984695
CrossRef - Qian L, Du M, Yang X, et al. Microanalysis Characterization and Immunomodulatory Effect for Selenium-Enriched Polysaccharide from Morchella esculenta (L.) Pers. Molecules. 2023;28(7):2885. doi:10.3390/molecules28072885
CrossRef - Xiaoying M, Zhiming H, Tao Y, et al. Elucidating the molecular mechanisms underlying anti-inflammatory effects of Morchella esculenta in the arachidonic acid metabolic pathway by network pharmacology and molecular docking. Scientific Reports. 2023;13(1). doi:10.1038/s41598-023-42658-1
CrossRef - Li S, Sang Y, Zhu D, Yang Y, Lei Z, Zhang Z. Optimization of fermentation conditions for crude polysaccharides by Morchella esculenta using soybean curd residue. Industrial Crops and Products. 2013;50:666-672. doi:10.1016/j.indcrop.2013.07.034
CrossRef - Liu C, Sun Y, Mao Q, et al. Characteristics and Antitumor Activity of Morchella esculenta Polysaccharide Extracted by Pulsed Electric Field. International Journal of Molecular Sciences. 2016;17(6):986. doi:10.3390/ijms17060986
CrossRef - Li Y, Chen H, Zhang X. Cultivation, nutritional value, bioactive compounds of morels, and their health benefits: A systematic review. Frontiers in Nutrition. 2023;10. doi:10.3389/fnut.2023.1159029
CrossRef - Wang D, Yin Z, Ma L, et al. Polysaccharide MCP extracted from Morchella esculenta reduces atherosclerosis in LDLR-deficient mice. Food & Function. 2021;12(11):4842-4854. doi:10.1039/d0fo03475d
CrossRef - Li Y, Yuan Y, Lei L, et al. Carboxymethylation of polysaccharide from Morchella angusticepes Peck enhances its cholesterol-lowering activity in rats. Carbohydrate Polymers. 2017;172:85-92. doi:10.1016/j.carbpol.2017.05.033
CrossRef - Chang, S. T. Cultivated mushroom. In: Hand Book of Applied Mycology,Foods and Feeds Vol. 3 Eds by Arora, D. K., Mukerji, K. G. and Marth, E. H., Marcel Dekker, Inc., New York. 1991;pp. 221–240.
- Tietel Z, Masaphy S. True morels (Morchella)—nutritional and phytochemical composition, health benefits and flavor: A review. Critical Reviews in Food Science and Nutrition. 2018;58(11):1888-1901. doi:10.1080/10408398.2017.1285269
CrossRef - Liu W, He P, Shi X, Zhang Y, Perez-Moreno J, Yu F. Large-Scale field cultivation of morchella and relevance of basic knowledge for its steady production. Journal of Fungi. 2023;9(8):855. doi:10.3390/jof9080855
CrossRef - Liu W., Cai Y.L., He P.X., et al. Artifical cultivation of Morchella. Compilation of information from scene view of morel cultivation and 2016 annual meeting of edible fungi association of Hubei Province; Chinese. 2016;pp. 27–36.
- Liu S.L., Li K.B., Zhu H., et al. The current situation of Morchella artificial cultivation technology and problem analysis. Edible Med Mushrooms (Chinese). 2016;24: 290–293.
- Qin X.B., Zhang G.Z., Shi X.D., inventors; Wang ZX, assignee et al. Morchella nutrition formula, nutrition bag, preparation method of nutrition bag and Morchella culture method. Chinese patent CN 105,191,667 A. 30 Dec, 2015.
- Shi D.Y., inventor; Chendu Tianlv Fungi Co Ltd, assignee. Preparation method of culture material formula and nutrition pack formula for morel. Chinese patent CN 103,819,281 A. 2014.
- Cotter, T. Organic mushroom farming and mycoremediation: simple to advanced and experimental techniques for indoor and outdoor cultivation. Vermont: Chelsea Green Publishing. 2014; p. 212–227.
- Xu Y, Tang J, Wang Y, et al. Large-scale commercial cultivation of morels: current state and perspectives. Applied Microbiology and Biotechnology. 2022;106(12):4401-4412. doi:10.1007/s00253-022-12012-y
CrossRef - Doctor V. Gucchi: wild mushrooms from Himalayas worth their weight in gold. The Economic Times, Mar 17, 2013 URL- https://economictimes.indiatimes.com/gucchi-wild-mushrooms-from-himalayas-worth-their-weight-in-gold/articleshow/19007096.cms
- Xu L, Zhang Y, Li H, Li J, Xu J. Challenges and Strategies for Continuous Cropping of Morchella: A Review. Horticulturae. 2024;10(12):1288. doi:10.3390/horticulturae10121288
CrossRef - Tan H, Kohler A, Miao R, et al. Multi‐omic analyses of exogenous nutrient bag decomposition by the black morel Morchella importuna reveal sustained carbon acquisition and transferring. Environmental Microbiology. 2019;21(10):3909-3926. doi:10.1111/1462-2920.14741
CrossRef - Tan H, Yu Y, Tang J, et al. Build your own mushroom soil: microbiota succession and nutritional accumulation in Semi-Synthetic substratum drive the fructification of a Soil-Saprotrophic morel. Frontiers in Microbiology. 2021;12. doi:10.3389/fmicb.2021.656656
CrossRef - Zhang Y, Sun S, Luo D, et al. Decline in Morel Production upon Continuous Cropping Is Related to Changes in Soil Mycobiome. Journal of Fungi. 2023;9(4):492. doi:10.3390/jof9040492
CrossRef - Wei-Ye L, Hong-Bo G, Ke-Xin B, Alekseevna SL, Xiao-Jian Q, Xiao-Dan Y. Determining why continuous cropping reduces the production of the morel Morchella sextelata. Frontiers in Microbiology. 2022;13. doi:10.3389/fmicb.2022.903983
CrossRef - Yin Q, Chen Z, He P, Liu W, Zhang W, Cao X. Allelopathic effects of phenolic acid extracts on Morchella mushrooms, pathogenic fungus, and soil-dominant fungus uncover the mechanism of morel continuous cropping obstacle. Archives of Microbiology. 2024;206(1). doi:10.1007/s00203-023-03790-8
CrossRef - Yu FM, Jayawardena RS, Thongklang N, Lv ML, Zhu XT, Zhao Q. Morel Production Associated with Soil Nitrogen-Fixing and Nitrifying Microorganisms. Journal of Fungi. 2022;8(3):299. doi:10.3390/jof8030299
CrossRef - Zou P, Guo Y, Ding S, et al. Autotoxicity of Endogenous Organic Acid Stress in Two Ganoderma lucidum Cultivars. Molecules. 2022;27(19):6734. doi:10.3390/molecules27196734
CrossRef - Duan M, Yang C, Bao L, et al. Morchella esculenta cultivation in fallow paddy fields and drylands affects the diversity of soil bacteria and soil chemical properties. Frontiers in Genetics. 2023;14. doi:10.3389/fgene.2023.1251695
CrossRef - Jalli M, Huusela E, Jalli H, et al. Effects of crop rotation on spring wheat yield and pest occurrence in different tillage systems: A Multi-Year Experiment in Finnish Growing conditions. Frontiers in Sustainable Food Systems. 2021;5. doi:10.3389/fsufs.2021.647335
CrossRef - Li C, Stomph TJ, Makowski D, et al. The productive performance of intercropping. Proceedings of the National Academy of Sciences. 2023;120(2). doi:10.1073/pnas.2201886120
CrossRef - Wang X, Duan Y, Zhang J, et al. Response of potato yield, soil chemical and microbial properties to different rotation sequences of green manure-potato cropping in North China. Soil and Tillage Research. 2022;217:105273. doi:10.1016/j.still.2021.105273
CrossRef - Momma N. Biological soil disinfestation (BSD) of soilborne pathogens and its possible mechanisms. Japan Agricultural Research Quarterly JARQ. 2008;42(1):7-12. doi:10.6090/jarq.42.7
CrossRef - Du XH, Yang ZL. Mating systems in true morels (Morchella). Microbiology and Molecular Biology Reviews. 2021;85(3). doi:10.1128/mmbr.00220-20
CrossRef - Sugano SS, Suzuki H, Shimokita E, et al. Genome editing in the mushroom-forming basidiomycete Coprinopsis cinerea, optimized by a high-throughput transformation system. Scientific Reports. 2017;7(1). doi:10.1038/s41598-017-00883-5
CrossRef - Kaminsky LM, Trexler RV, Malik RJ, Hockett KL, Bell TH. The inherent conflicts in developing soil microbial inoculants. Trends in Biotechnology. 2019;37(2):140-151. doi:10.1016/j.tibtech.2018.11.011
CrossRef - O’Callaghan M, Ballard RA, Wright D. Soil microbial inoculants for sustainable agriculture: Limitations and opportunities. Soil Use and Management. 2022;38(3):1340-1369. doi:10.1111/sum.12811
CrossRef

