Introduction
Broccoli (Brassica oleracea var. italica Plenck), a Brassicaceae family member, is becoming more and more popular worldwide because of its many health advantages and is grown in a broad variety of cultivars. Broccoli is believed to be the earliest member of the Cole crops, which emerged from the wild varieties of cabbage or kale. It is well-known throughout the Terai zone for its tender knob and processed foods like soup and vegetable curry preparation. It contains a number of bioactive chemicals that boost immunity and lower cholesterol, cardiovascular disease, and some types of cancer.1 Since people are becoming more concerned about their health, there has been a tremendous increase in demand for this product because of its extremely high nutritional value. There has been conjecture that farmers who grow broccoli, a highly prized crop, may earn more financial returns because of the climate in West Bengal’s Terai region of India. Accurate and consistent seed germination, along with rapid seedling growth, is a crucial element in time-synchronized crop establishment. Stresses that arise between seeding and seedling establishment are especially harmful to seeds.2 Through reduced water intake, hormone imbalances, lowering antioxidant levels and cellular damage, drought stress limits plant germination and seedling vigour.3-5 The kind of plant tissue, the amount of stress, and the duration of stress all influence the extent of growth reduction.6 Several studies have shown that when plants are subjected to osmotic stress, their growth is stunted.7,8 Plants are most frequently subjected to artificial osmotic stress via the use of polyethylene glycol (PEG). It has been discovered to negatively impact the germination and growth of broccoli. Broccoli’s typical emergence, development, biochemical characteristics, and yield are all destroyed by osmotic stress. To counteract these negative impacts, various practical mitigation approaches are typically implemented to bolster a plant’s resilience to stress. Keeping this in mind, it could be beneficial to choose a superior cultivar to utilize it in the prevailing drought stress conditions in the natural environment.
This study mainly focuses on eleven broccoli cultivars that are predominantly cultivated in West Bengal, India and primarily aims to examine the impact of osmotic stress caused by polyethylene glycol on broccoli seed germination and seedling vigour, as well as to identify the best-performing cultivar for cultivation by indigenous farmers. The local farmers will surely benefit from this work because, it should be pointed out, none of these eleven common broccoli cultivars from the Terai region—which has a comparatively warmer climate—have ever been screened together under drought stress.
Materials and Methods
Experimental set up
Eleven common certified Broccoli cultivars (FANTASY F1, GREEN MAGIC, GAMORA, GREEN VELVET, DIANA, SAMAYRA, PARAISO, LUCKY F1, SAKI, SEAN, TITANIUM) were collected and employed to examine the effect of osmotic stress induced by PEG 6000 on their germination and early seedling growth. The experiment was performed in petriplates using absorbant cotton and blotting paper. The seeds, selected for their uniform size, were subjected to surface sterilization in a 1% (v/v) sodium hypochlorite solution for five minutes. Thereafter, they were meticulously rinsed twice with double-distilled water. Sixteen seeds from each cultivar were positioned in petriplates containing the designated PEG 6000 concentrations (0, -0.4, -0.8, and -1.2 MPa) on the basis of entirely randomized design with 3 replication per treatment. Every day, an appropriate PEG 6000 solution was added to each petriplate. The quantity of germinated seeds was recorded daily over a duration of 7 days. Seedling vigour was assessed after 14 days by measuring the root length (RL) and shoot length (SL) in centimeters. To finalize the investigation, several apparatuses including an electronic balance, a petriplate, a beaker, absorbent cotton, blotting paper, and a micropipette were employed. The various germination parameters, stress tolerance indices, seedling vigour indices and phytotoxicity indices were determined using the following formulae:
Analysis of Germination attributes. Following formulae were used to study germination parameters.
Total Germination Percentage (TG %)=(Total number of germinated Seeds/Total Seeds)x100.9
Germination Index (GI) = (7 x n1) + (6 x n2) + … + (1 x n7).10
Where, n1, n2, … n7 are the number of germinated seeds on the first, second, … … the seventh day and 7, 6, … 1 are the weights given to the number of germinated seeds on the first, second, … … the seventh day, respectively.
Mean Germination Time (MGT) = ∑ (n × d)/N.11
Where, n represents the number of seeds germinated each day, d denotes the number of days since the investigation commenced, and N signifies the total number of seeds germinated at the end of the experiment.
Speed of Germination (SG) = (n1/d1) + (n2/d2) + (n3/d3) + … + (n7/d7).12
Where, n represents the quantity of germinated seeds at each time interval and d denotes the number of days since the start of planting.
Coefficient of Velocity of Germination (CV) = ((∑NiTi) /(∑Ni))*100.13
Where, Ni indicates the number of seeds germinated on the ith day and Ti denotes the duration from the commencement of the experiment to the ith interval.
Mean Germination Rate (MGR) = 1 / MGT.14
Reduction of Germination (RPG) = % germination of control – germination of treatment / % germination of control x 100.15
Stress tolerance analysis.
Promptness Index (PI) = nd2 (1.00) + nd4 (0.75) + nd6 (0.5) + nd8 (0.25).16
Where, n denotes number of germinated seeds on day d.
Germination Stress tolerance Index (GSI) = (P.I of stressed seeds / P.I control seeds) × 100.16
RL Stress Tolerance Index (RLSI) = (RL of stressed plant/RL of control plants) × 100.16
Plant Height Stress Tolerance Index (PHSI) = (Height of stressed plant / Height of control plants) × 100.16
Shoot Height Stress Tolerance Index (SHSI)=(SL of stressed plant/SL of control plant)×100.16
Seedling Vigour and Phytotoxicity analysis
Vigour Index (VI) = (RL + SL) x germination percentage of seed.17
Root phytotoxicity Percentage (RPP)=(RL of Control–RL of treatment)/RL of Control×100.18
Shoot phytotoxicity Percentage (SPP)=(SL of Control–SL of treatment)/SL of Control×100.18
All the graphs were made with the help of OriginLab software (https://www.originlab.com › origin). Data were analyzed using Duncan’s Multiple Range Test (DMRT) at p>0.05.
Results
Total Germination Percentage (TG %).
GREEN MAGIC, GREEN VELVET, and PARAISO showed the highest values (all with identical values) under control in the case of TG %, a measure of seed viability that indicates seed quality and vigour (Figure 1). However, GREEN VELVET and PARAISO did not show consistent results at higher stress concentrations of -0.8 and -1.2 MPa. Similarly, GREEN MAGIC exhibited the highest TG% values (68% and 14% respectively) among all the cultivars at elevated stress concentrations of -0.8 and -1.2 MPa. Conversely, the TITANIUM cultivar demonstrated the lowest TG% value across each treatment, signifying poor performance compared to all other cultivars. Notably, under control treatment, majority of the cultivars showed similar results with slight variations, but quite significant variations in TG% values were displayed by the cultivars at the elevated stress concentrations of -0.8 and -1.2 MPa.
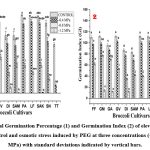 |
Figure (1-2): Total Germination Percentage (1) and Germination Index (2) of eleven Broccoli seeds grown under control and osmotic stress induced by PEG at three concentrations (-0.4, -0.8, and -1.2 MPa) with standard deviations indicated by vertical bars. |
Germination Index (GI)
In this experiment, the highest values of GI (Figure 2) were recorded in GREEN MAGIC, GREEN VELVET, and PARAISO showing identical values under control set. However, at elevated stress concentration (-0.8 MPa and -1.2 MPa), only GREEN MAGIC consistently demonstrated superior results compared to the other cultivars. As a matter of fact, few cultivars like GREEN VELVET, LUCKY F1, and SAKI showed marginally higher GI values than that of GREEN MAGIC at lower stress concentration (-0.4 MPa) though their results were not satisfactory at elevated stress concentrations (-0.8 MPa, and -1.2 MPa). For GI values at the highest stress concentration of -1.2 MPa, after GREEN MAGIC only GAMORA and DIANA showed good results; performance of all other cultivars were not remarkable. Among all cultivars, the TITANIUM cultivar had the lowest GI values both under control and stressed concentrations.
Mean Germination Time (MGT)
The results indicated that the maximum value of MGT (Figure 3) was recorded in DIANA immediately followed by SAMAYRA under control condition indicating the slower germination compared to other cultivars. Under the lowest stress condition (-0.4 MPa), TITANIUM exhibited the highest MGT value compared to other cultivars. Under a stress concentration of -0.8 MPa, no seeds germinated in TITANIUM. Likewise, at a concentration of -1.2 MPa, only 4 cultivars (GREEN MAGIC, GAMORA, DIANA and SAKI) showed significant results for MGT while remaining seven cultivars (FANTASY F1, GREEN VELVET, SAMAYRA, PARAISO, LUCKY F1, SEAN, TITANIUM) showed no germination. On the contrary, the control seeds of GREEN MAGIC, GREEN VELVET, and PARAISO exhibited the lowest (same) MGT value. At -0.8 MPa stress levels, GREEN MAGIC exhibited the lowest MGT, succeeded by GREEN VELVET and FANTASY F1, respectively.
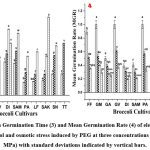 |
Figure (3-4): Mean Germination Time (3) and Mean Germination Rate (4) of eleven Broccoli seeds grown under control and osmotic stress induced by PEG at three concentrations (-0.4, -0.8, and -1.2 MPa) with standard deviations indicated by vertical bars. |
Mean Germination Rate (MGR)
In this experiment, under control conditions, the highest value of MGR (Figure 4) was recorded in GREEN MAGIC, GREEN VELVET, and PARAISO, having identical values. Though under the lowest stressed concentration (-0.4 MPa) GREEN MAGIC displayed moderately higher MGR value, under -0.8 and -1.2 MPa stress concentrations GREEN MAGIC displayed the highest MGR value. At the highest osmotic stress concentration (-1.2 MPa), apart from GREEN MAGIC, only three cultivars, namely GAMORA, DIANA, and SAKI, exhibited significant results, whereas no notable MGR values were recorded for the remaining seven cultivars: GREEN VELVET, FANTASY F1, PARAISO, LUCKY F1, SAMAYRA, SEAN, and TITANIUM.
Speed of Germination (SG)
In this experiment, the highest SG values (Figure 5) were recorded in GREEN MAGIC, GREEN VELVET, and PARAISO showing identical values under control group. At the lowest concentration (-0.4 MPa), the highest value of SG was observed in SAKI. However, at elevated stress concentrations (-0.8 MPa and -1.2 MPa), only GREEN MAGIC exhibited superior outcomes to all the other cultivars. At the highest stress concentration of -1.2 MPa, GAMORA, DIANA and SAKI showed meager results for SG. However, in the case of TITANIUM, the SG values were consistently lower across all concentrations.
Coefficient of Velocity of Germination (CV)
DIANA obtained the highest CV values (Figure 6) in both the control and -0.8 MPa conditions. SAKI and TITANIUM had the highest CV values, at -1.2 and -0.4 MPa, respectively. GREEN MAGIC’s CV values were moderate. Notably, larger CV values were the outcome of germination observed in subsequent days. The GREEN MAGIC seeds germinated very rapidly. At the greatest stress level of -1.2 MPa, only DIANA, GAMORA, and GREEN MAGIC demonstrated noteworthy results; all other CV values for remaining cultivars were zero.
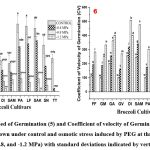 |
Figure (5-6): Speed of Germination (5) and Coefficient of velocity of Germination (6) of eleven Broccoli seeds grown under control and osmotic stress induced by PEG at three concentrations (-0.4, -0.8, and -1.2 MPa) with standard deviations indicated by vertical bars. |
Reduction of Germination (RPG)
The results indicated that, in lower stress concentrations (-0.4 MPa and -0.8 MPa), TITANIUM exhibited the highest value of RPG (Figure 7). In the highest stress concentrations (-1.2 MPa), TITANIUM, SEAN, LUCKY F1, PARAISO, and GREEN VELVET exhibited the highest RPG values. However, at elevated stress concentrations of -0.8 MPa and -1.2 MPa, only GREEN MAGIC exhibited the lowest value of RPG indicating greater seed viability.
Germination Stress tolerance Index (GSI)
In case of all stressed concentration, GREEN MAGIC had the highest GSI value, while TITANIUM demonstrated the lowest GSI value (Figure 8) compared to other cultivars. At the highest stress concentration of (-1.2 MPa), after GREEN MAGIC, only GAMORA and DIANA showed remarkable results, all others showing zero (non-significant) values.
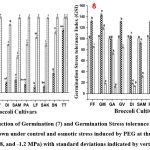 |
Figure (7-8): Reduction of Germination (7) and Germination Stress tolerance Index (8) of eleven Broccoli seeds grown under control and osmotic stress induced by PEG at three concentrations (-0.4, -0.8, and -1.2 MPa) with standard deviations indicated by vertical bars. |
Root Length Stress Tolerance Index (RLSI)
In this experiment, the maximum values of RLSI (Figure 9) were recorded under control conditions for all selected cultivars. At the lower stress concentration of (-0.4 MPa), SAKI (123.16) exhibited the highest values of RLSI followed by GREEN MAGIC (110.71), while at the elevated stress concentration (-0.8 MPa), GREEN MAGIC exhibited the highest value (82.57) followed by GAMORA (67.52). However, TITANIUM demonstrated the lowest RLSI value (zero) in higher stress concentrations.
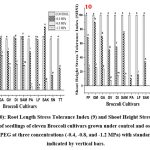 |
Figure (9-10): Root Length Stress Tolerance Index (9) and Shoot Height Stress Tolerance Index (10) of seedlings of eleven Broccoli cultivars grown under control and osmotic stress induced by PEG at three concentrations (-0.4, -0.8, and -1.2 MPa) with standard deviations indicated by vertical bars. |
Shoot Height Stress Tolerance Index (SHSI) and Plant Height Stress Tolerance Index (PHSI).
The results demonstrated that at stress concentrations of -0.4 MPa and -0.8 MPa, GREEN MAGIC exhibited the greatest values of SHSI (Figure 10) and PHSI (Figure 11), outperforming all other cultivars. The minimum values for both SHSI and PHSI were recorded in TITANIUM. At the maximal stress concentration of (-1.2 MPa), all cultivars demonstrated no noteworthy outcomes.
Vigour Index (VI)
In the context of VI, GREEN VELVET exhibited the highest value in the control group followed by LUCKY F1 (Figure 12). However, under stress conditions they failed to show consistently good results. On the contrary, in lower stress concentrations (-0.4 MPa and -0.8 MPa), only GREEN MAGIC exhibited the highest values of VI, specifically 1065.54 and 486.75 respectively. TITANIUM consistently demonstrated the poorest results with lowest VI values both in control as well as under stress conditions. Notably, the VI results for all cultivars were insignificant under the higher stress condition (-1.2 MPa). This is because, even though some seeds germinated, they failed to grow into a seedling, resulting in a vigour index of zero.
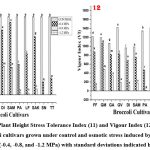 |
Figure (11-12): Plant Height Stress Tolerance Index (11) and Vigour Index (12) of seedlings of eleven Broccoli cultivars grown under control and osmotic stress induced by PEG at three concentrations (-0.4, -0.8, and -1.2 MPa) with standard deviations indicated by vertical bars. |
Root phytotoxicity Percentage (RPP) and Shoot phytotoxicity Percentage (SPP).
The results indicated that the highest level of RPP (Figure 13) were obtained in SEAN at lower stress concentration (-0.4 MPa), whereas GREEN MAGIC demonstrated the lowest RPP at lower stress concentrations of -0.4 MPa and -0.8 MPa (0.00 and 17.429 respectively).
In the context of SPP, TITANIUM exhibited the highest value at lower stress concentration (-0.4 MPa). On the contrary, in lower stress concentrations (-0.4 MPa and -0.8 MPa), only GREEN MAGIC shows the lowest values of SPP (Figure 14), specifically 10.20 and 74.00 respectively. At the maximum stress concentration (-1.2 MPa), all cultivars exhibited values of 100% or near about 100% showing inhibition of growth as we know that a higher RPP and SPP value indicate greater level of phytotoxicity and more harmful to root and shoot growth respectively.
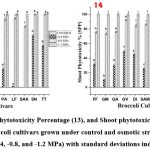 |
Figure (13-14): Root phytotoxicity Percentage (13), and Shoot phytotoxicity Percentage (14) of seedlings of eleven Broccoli cultivars grown under control and osmotic stress induced by PEG at three concentrations (-0.4, -0.8, and -1.2 MPa) with standard deviations indicated by vertical bars. |
Discussion
In the present study, the elevated PEG concentrations decreased the germination rate and seedling vigour indices (like TG%, GI, MGT, MGR, SG, CV and VI) in relation to the control across all broccoli cultivars. This result is in agreement with previous works in multiple crops.19 Under extreme osmotic stress the noted decline in shoot and root development corroborates earlier research demonstrating that drought stress adversely affects morphological and physiological characteristics throughout the seedling phase of broccoli. This may result from hindered water absorption and diminished enzyme activity essential for germination.20 The lowest values recorded for RPG at osmotic stress of -0.8 and -1.2 MPa, respectively, unequivocally demonstrate accelerated germination and enhanced seed viability in GREEN MAGIC in comparison to other cultivars. Results indicated that only GREEN MAGIC consistently exhibited higher performance relative to other cultivars across many important morphological parameters especially TG%, SG, MGR, RPG, GSI, and GI. Only GREEN MAGIC exhibited the highest values of VI relative to other cultivars at lowered stress concentrations (-0.4 MPa and -0.8 MPa). This indicates that GREEN MAGIC possesses enhanced stress tolerance and a higher potential for crop output, whereas TITANIUM consistently demonstrated the least favorable results, exhibiting the lowest VI values in both control and stress conditions. This indicates poor performance that may require additional scrutiny under the specific stress conditions. Nonetheless, occasionally one or another cultivar might outperform GREEN MAGIC in any particular treatment, but except GREEN MAGIC overall consistent results for these cultivars were not achieved under varying drought stress conditions caused by PEG. Furthermore, an elevated index value of SHSI and PHSI in GREEN MAGIC indicates that the cultivar’s height is minimally diminished, reflecting a high degree of tolerance. Conversely, a low index value of SHSI and PHSI in some cultivars, particularly TITANIUM and SEAN, indicates that the stress resulted in a substantial decrease in height, signifying susceptibility. Similarly, lowest values obtained for RPP and SPP in GREEN MAGIC signifies a less toxic impact on the plant’s roots or shoots, showing less damage and enhanced vigour. On the contrary, a significant percentage of RPP and SPP in SEAN and TITANIUM would suggest major detriment, including stunted growth, foliar damage, or potential plant mortality, due to exposure to stress conditions.
Conclusion
The results clearly exhibited GREEN MAGIC’s superiority compared to other cultivars under various drought stress conditions while TITANIUM displayed the least favorable results, identifying it as the most inferior cultivar under such conditions. So, GREEN MAGIC should be preferred for cultivation among the eleven common cultivars. The findings of this study will definitely be beneficial for local farmers’ agriculture for the utilization of the chosen broccoli cultivar under drought stress conditions. We look forward to conducting additional research on this resilient broccoli cultivar to improve seedling vigour and yield after priming with various appropriate elicitors to mitigate the negative effects of drought stress, as well as addressing the genetic and physiological processes of drought resistance in agricultural contexts.
Acknowledgement
The authors express their heartfelt gratitude for the help extended by Late (Dr.) Palash Mandal, Associate Professor, Department of Botany, University of North Bengal (NBU) and Dr. Gyan Chandra Pariyar, Assistant Professor, Department of Food Technology, NBU.
Funding Sources
The author(s) did not receive any financial assistance for the research, authoring, or publishing of this work.
Conflict of Interest
The authors do not have any conflicts of interest.
Data Availability Statement
All of the datasets created or analyzed during this research work are included in the publication.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Papiya Dey: Investigation, Formal Analysis, Data Curation, Methodology, and Conceptualization.
Sujoy Kumar Sen: Editing, Reviewing, and Supervision.
References
- Syed R. U., Moni S. S., Break M. K. B., et al. Broccoli: A multi-faceted vegetable for health: An in-depth review of its nutritional attributes, antimicrobial abilities, and anti-inflammatory properties. Antibiot. 2023;12(7):1157.
CrossRef - Carter L. M., Chesson J. H. Two USDA researchers develop a moisture seeking attachment for crop seeders that is designed to help growers plant seed in soil sufficiently moist for germination. Seed World. 1996;134:14-15.
- Khan A. A. Preplant physiological seed conditioning. Hortic Rev. 2010;13:131-181.
CrossRef - Flowers T. J. Improving crop salt tolerance. Exp. Bot. 2004;55(396):307-319. https://doi.org/10.1093/ jxb/erh003
CrossRef - Ashraf M., Akram N. A., Arteca R. N., Foolad M. R. The physiological, biochemical and molecular roles of brassinosteroids and salicylic acid in plant processes and salt tolerance. Critical Reviews. Plant Sci. J. 2010;29(3):162-192. https://doi.org/10.1080/07352689.2010.483580
CrossRef - Meloni D. A., Oliva M. A., Martinez C. A., Cambraia J. Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Exp. Bot. 2003;49(1):69-76. https://doi.org/10.1016/S0098-8472(02)00058-8
CrossRef - Munns R., Tester M. Mechanisms of salinity tolerance. Rev. Plant Biol. 2008; 59(1):651-681. https://doi.org/10.1146/annurev.arplant.59.032607.092911
CrossRef - Taiz L., Zeiger E. Plant Physiology, Sunderland, Massachussetts, Sinauer Associates. 4th USA: Publishers; 2006.
- Faijunnahar M., Baque A., Habib M. A., Hossain H. M. M. T. Polyethylene glycol (PEG) induced changes in germination, seedling growth and water relation behavior of wheat (Triticum aestivum) genotypes. Univers J Plant Sci. 2017;5(4):49-57.
CrossRef - Kusmiyati F., Sas M. G. A., Herwibawa B. Mutagenic effects of gamma rays on soybean (Glycine max) germination and seedlings. InIOP Conference Series: Earth and Environ. Sci. 2018;102(1):022059. DOI 10.1088/1755-1315/102/1/012059
CrossRef - Ellis R. H., Roberts E. H. The quantification of ageing and survival in orthodox seeds. Seed Sci. Technol. 1981;9:373-409.
- Ruan S., Xue Q., Thlkowska K. Effect of seed priming on germination and health of rice (Oryza sativa) seeds. Seed Sci. Technol. 2002;30:451-458.
- Moghadam P. A., Alaei Y. Evaluation of important germination traits of soybean genotypes through factor analysis in osmotic drought stress conditions. Env. Pharmacol. Life Sci. 2014;3(5):05-08.
- Ranal M. A., Santana D. G. D., Ferreira W. R., Mendes-Rodrigues C. Calculating germination measurements and organizing spreadsheets. J. Bot. 2009;32:849-855. https://doi.org/10.1590/S0100-84042009000400022
CrossRef - El-Madidi S., El-Baroudi B., Aameur F. B. Effects of salinity on germination and early growth of barley (Hordeum vulgare) cultivars. Int. J. Agric. Biol. 2004;6:767-770.
- Ashraf M. Y., Akhtar K., Hussain F., Iqbal J. Screening of different accessions of three potential grass species from Cholistan desert for salt tolerance. J. Bot. 2006; 38(5):1589-1597.
- Islam A. A., Anuar N., Yaakob Z., Ghani J. A., Osman M. Combining ability for germination traits in Jatropha curcasSci. World J. The. 2013;(1):935981. https://doi.org/10.1155/2013/935981
CrossRef - Asmare H. A., Ambo E. Impact of salinity on tolerance, vigour, and seedling relative water content of haricot bean (Phaseolus vulgaris) cultivars. J. Plant Sci. 2013; 1(3):22-27.
- Sani D. O., Boureima M. M. Effect of polyethylene glycol (PEG) 6000 on germination and seedling growth of pearl millet [Pennisetum glaucum (L.) R. Br.] and LD50 for in vitro screening for drought tolerance. J. Biotechnol. 2014;13(37). DOI: 10.5897/AJB2013.13514
CrossRef - Rajani V., Ramesh K., Anamika N. Drought Resistance Mechanism and Adaptation to Water Stress in Sorghum [Sorghum bicolor (L.) Moench]. J. Bioresour. 2018; 9(1):167-172.
CrossRef
Abbreviation
FF: FANTASY F1
GM: GREEN MAGIC
GA: GAMORA
GV: GREEN VELVET
DI: DIANA
SAM: SAMAYRA
PA: PARAISO
LF: LUCKY F1
SAK: SAKI
SN: SEAN
TT: TITANIUM

