Introduction
Anthropogenic activities and global warming have increased drought, which creates a serious challenge to worldwide food production.1 Drought stress is a major abiotic factor that negatively impacts crop productivity, varying from brief and mild episodes to severe and extended conditions that significantly restricting crop yields.2,3 Water stress impacts a plant’s water potential and turgor pressure, disrupting normal cellular functions and leading to significant changes in its morphological and physiological characters.4,5
Water content is a key factor influencing plant growth and is largely impacted by drought conditions and the level of nutrients present in the soil. The transport and diffusion of water-soluble nutrients, carried out by water through the roots, are also impacted.6–8 The drought stress in plants leads to an increased production of free radicals, which triggers oxidative damage such as lipid peroxidation, degradation of cellular membranes.9–11 Such physiological impairments may drastically reduce crop productivity, with yield losses varying between 30% and 90% based on the plant species and the specific growth stage exposed to drought stress.12
In recent times, to keep up with the increasing demand for food and enhance agricultural productivity, a wide range of chemical pesticides and fertilizers have been employed.13,14 However, the intensive use of chemicals can cause significant environmental damage, posing major ecological threats and creating serious risks to human health.15,16
Current strategies to enhance plant growth and productivity include traditional breeding techniques, genetic engineering, and priming approaches.17–20 While these methods have shown considerable promise, each comes with its own set of challenges and constraints, ranging from lengthy development times and regulatory hurdles to inconsistent results under field environment.21,22 Nowadays, Plant Growth-Promoting Rhizobacteria (PGPR) have arisen as a fascinating and effective alternative to traditional inputs, gaining recognition as biofertilizers in modern agriculture.23 PGPRs are a diverse group of microorganisms includes Bacillus, Pseudomonas, Azospirillum, Azotobacter, Klebsiella, Enterobacter, Alcaligenes, Arthrobacter, Burkholderia, and Serratia.24 They display host specificity, indicating that their effectiveness can vary across different plant species or cultivars.25 Selecting the appropriate species of PGPR tailored to each plant type is essential to maximize their positive impacts on plant growth and resilience to stress. Only the identified species can then be artificially inoculated in the rhizosphere of that plant along with the existing flora.
Inoculating plants with PGPR in the rhizospheric soil is one solution to counter the problem of drought stress for sustainable farming.26 They can enhance plant development by either producing beneficial phytohormones—like gibberellins, auxins, and cytokinins—or by facilitating improved nutrient acquisition from the soil, including nitrogen, phosphorus, and essential micronutrients. These mechanisms not only boost root and shoot growth but also help plants better withstand environmental stresses. In addition to their direct growth-promoting effects, PGPR also support plant health indirectly by producing antagonistic compounds—such as antibiotics, siderophores, and hydrogen cyanide—that suppress harmful phytopathogens.27,28 Numerous studies suggest that bacterial strains can promote plant growth even in drought-stressed conditions.29–31 These bacterial strains enhance a plant’s drought tolerance, making them particularly beneficial for cultivation in arid and semi-arid regions.32
This study aims to isolate a PGPR strain from the rhizosphere of Celosia argentea, which can enhance plant growth and increase survival rates under drought conditions. The Celosia argentea is an ornamental plant belonging to the Amaranthaceae family. Celosia argentea shows fast growth, is strongly adaptable, and has a high propagation rate,33 but it is sensitive to drought stress. Drought significantly damages the cellular membrane of Celosia argentea,34 but the plant selected for this study was well adapted to drought conditions and thrived in a dry area, drawing significant attention.
Materials and Methods
Soil Sample
The rhizospheric and surrounding soil from Celosia argentea was collected from Shelgaon village in Barshi tehsil, Solapur district, India. The plant was uprooted, and the adhered soil was removed and collected in a sterile container. The soil sample was subjected to analysis for various parameters: pH, electrical conductivity, organic carbon, calcium, magnesium, chloride, carbonate, bicarbonate, and other metals using standard methods of the FAO (2020).35
Isolation of bacteria
The collected rhizospheric soil sample was inoculated in presterilized NB and then transferred to a nutrient agar plate. Morphologically distinct colonies were selected and transferred to nitrogen-free mineral agar plates with the following composition (gL⁻¹): glucose, 20.0; FeCl₃•6H₂O, 0.025; MgSO₄•7H₂O, 0.5; Na₂MoO₄•2H₂O, 0.005; CaCl₂, 0.05; K₂HPO₄, KH₂PO₄, 0.2; 0.8; agar, 15; pH 7.0. The inoculated petri-plates were incubated at 30°C for 2 days. Bacterial colonies showing growth on the nitrogen-free medium were selected for further study.
Biochemical and Morphological Characterisation
The bacterial strain was identified based on basis of morphological and biochemical properties, determined by observing the bacterial colony and cell size, shape, spore formation, and Gram characteristics using Gram staining under 100x oil immersion on a Micron Optic MONO CXL microscope. Several biochemical tests were performed following Bergey’s Manual of Determinative Bacteriology.
Identification of bacteria
The bacterial isolate was identified through its morphological and biochemical properties. Molecular identification was achieved by sequencing the 16S rDNA, which was carried out at the National Collection of Industrial Microorganisms, CSIR-National Chemical Laboratory, Pune, India. The obtained sequence was analysed against database sequences using BLASTn, accessible through the NCBI server. Sequences showing the maximum similarity as determined by the BLAST analysis were retrieved and utilized for constructing a phylogenetic tree using Molecular Evolutionary Genetics Analysis (MEGA11). Additionally, the nucleotide sequence of the isolate has been submitted to the GenBank database.
Assessment of bacterial drought stress resistance
The bacterial strain was isolated and subsequently transferred to a nutrient broth. Polyethylene Glycol (PEG) 8000 was added to the nutrient broth to mediate osmotic pressure. The concentrations of PEG 8000 used were as follows (in g/g): 0.023, 0.037, 0.042, 0.051, and 0.062. These concentrations corresponded to osmotic pressures of -0.15, -0.30, -0.36, -0.48, and -0.73 Mpa, respectively. According to Michel (1983),36 the osmotic pressure at a temperature 25°C was calculated using the equation given below:
![]()
In the 100 ml of the above-prepared stress medium, 2 ml of a 24-hour-old bacterial culture from the nutrient broth was inoculated and kept at 25°C for 2 days. After 2 days, bacterial growth was measured by the optical density at 600nm against uninoculated broth using a spectrophotometer (Bioera single beam spectrophotometer). The culture growth in normal conditions (without PEG 8000) was used for comparison.
ACC deaminase
The ACC deaminase-producing ability of an organism was confirmed as per protocol suggested by Patil et al. (2016),37 for this minimal ACC medium (in g/L, Glucose 2.0, Sodium citrate 2.0, CaCO3 4.0, C6H11KO7 2.0) with final concentration of ACC was 3.0 mmol l-1, prepared as per Penrose and Glick (2003),38 containing 0.005% of bromothymol blue. After 24 hours of incubation at 30°C, the colony developed a blue colouration, suggesting its potential to produce ACC deaminase. This method offers only a qualitative assessment of ACC deaminase activity.
Phosphate solubilization index
The bacterial strain selected based on drought tolerance was evaluated for phosphate solubilization index. A bacterial colony from Nutrient agar was inoculated on a Pikovskayas agar plate and incubated at 30°C for 3 days. Following incubation, the diameters of both the colony and the zone were recorded, and the phosphate solubilization index was calculated with the formula below.39
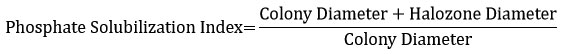
Siderophore production and nitrogen-fixing ability
The siderophore production of the bacterial strain was performed and analysed by spot inoculation of an overnight-grown culture on Chrome Azurol-S agar plates, and the plates were incubated for 2 days at 30 °C.40
The nitrogen-fixing ability of the selected bacterial strain was analyzed by inoculating it on nitrogen-free Jensen’s medium with composition g/L: Sucrose 20, MgSO4 0.5, NaCl 0.5, K2HPO4 1, FeSO4 0.1, Na2MoO4 0.005, CaCO₃ 2, Agar 15.
HCN and Ammonia Production
HCN production test was done by using a Nutrient agar medium with glycine (4.4 g/L). The isolated colony of the strain was inoculated on the Nutrient agar plate. A Whatman No. 1 filter paper saturated with picric acid (0.5%) and sodium carbonate (2%) solution was placed on the inner surface of the Petri dish lid, and the plates were incubated at 30 °C for 4 days.41 After incubation, a shift in the color of filter paper from yellow to orange, red, and brown corresponds to low, medium, and high levels of HCN production, respectively.42
The bacterial colony was inoculated in peptone broth g/L: 10.0 g peptone; 5.0 g NaCl; and 7.0 pH 43 incubated at 30°C for 2 days. Following incubation, Nessler’s reagent (0.5 ml ) was added, and observed for faint yellow, yellow, and brown colours, indicating the lowest to high production of ammonia.44
Indole‑acetic acid
The selective bacterial strains’ ability to secrete Indole‑acetic acid was analyzed by inoculating a colony in 100 ml Luria broth with 10 mg of tryptophan. After incubation, the broth was centrifuged at 10733 X G. The 1 ml of supernatant was added with 2 ml of Salkowski’s reagent (2% 0.5 FeCl3 in 35% HClO4), and the reaction mixture was incubated in dark for 30 min. The absorbance was recorded at 530nm. All the above-mentioned microbiological tests were performed in triplicate.
Seed germination study
The seed germination study was performed in vitro using the Petri plate method with sterilized blotting paper, and each experiment was carried out in three replicates to ensure accuracy and reproducibility. To eliminate contaminants, wheat seeds were treated with 0.1% mercuric chloride for five minutes, then thoroughly rinsed with double-distilled water to ensure effective surface sterilization. For biopriming, the seeds were immersed in a bacterial suspension containing 10⁷ CFU/mL for 2 hours, whereas control seeds were soaked in distilled water under identical conditions. The bacterial suspension was prepared by inoculating 5 μL of bacterial culture into 100 mL of nutrient broth, then incubating at 30°C for 24 hours to allow bacterial growth. Post-soaking, the seeds were placed onto sterilized blotting paper within Petri plates, moistened with 5 mL of either double-distilled water (control) or the bacterial suspension, as described by.45 Drought stress was artificially induced by adding 5 mL of a 5% PEG 8000 solution, equivalent to an osmotic potential of -0.47 MPa, into the Petri plates. Notably, the isolated bacterial strain exhibited robust growth under osmotic pressures as extreme as -0.48 MPa, demonstrating its adaptability.
Each Petri plate contained 15 seeds, and four distinct experimental groups were established: (1) water control, (2) bacterial control (bacterially primed seeds), (3) seeds treated with 5% PEG 8000 alone to simulate drought stress, and (4) seeds treated with both 5% PEG 8000 and bacterial suspension. The plates were kept at 25°C in a controlled growth chamber to assess germination under the specified conditions. Statistical analysis was done using MS-Excel software. Seed germination percentage was assessed after 4 days using the standard formula:
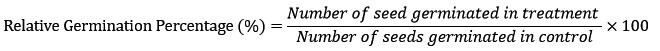
Results and Discussion
Soil sample and bacterial isolation
The rhizospheric and surrounding soil from Celosia argentea was collected from Shelgaon village of Solapur district (India) (Fig. 1). The district is considered drought-prone because of inadequate rainfall, with an annual average rainfall of 545mm. Solapur is one of 72 drought-prone districts in India.
 |
Figure 1: Map of the sample collection site |
The soil sample was collected and analyzed for different physical and chemical characteristics (Table 1). In the soil, a reservoir of nutrients is available and required for crop plants, but not at optimum levels. Soil analysis is needed to determine the level of available nutrients and soil quality for specific crop plants. Analysing soil can yield valuable insights that help optimise nutrient utilisation and enhance overall agricultural productivity. The sample was subjected to the isolation of drought stress-tolerant PGPR. The five morphologically distinct colonies were selected and inoculated on a nitrogen-free medium. Among five isolates, one that could fix nitrogen and was able to tolerate stress induced by 5% PEG 8000 (-47MPa) was selected for further study. The bacteria’s stress tolerance was verified by observing the optical density of their growth after incubation. For comparison, bacteria cultured in nutrient broth without PEG 8000 were the control (data not shown).
Table 1: Physicochemical analysis of soil sample
| Soil Analysis | Result |
| Calcium (mEq%) | 0.4 |
| Organic carbon (%) | 0.9 |
| pH | 7.08 |
|
EC (mili-siemens cm-1) |
0.885 |
| Magnesium (mEq%) | ND |
| Nitrogen kh-1 | 210 |
| Phosphate kh-1 | 4.3 |
|
Potassium kh-1 |
270 |
ND- Not Determined
Identification of bacterial strain
The isolated bacterium was characterized as Gram-positive, rod-shaped, motile, and endospore-forming. It exhibited growth in a pH range of 5 to 8, with optimal growth at pH 7. The temperature range for growth was 15°C to 40°C, with the optimal temperature at 30°C, and it was inhibited at 50°C. The bacterium tolerated salt concentrations up to 7.5%. Biochemical tests and 16S rDNA molecular identification confirmed its identity. The partial 16S rDNA sequence of the bacterial isolate was acquired from the National Collection of Industrial Microorganisms, CSIR-National Chemical Laboratory, Pune, India. Following its acquisition, the sequence was submitted to GenBank under the accession number PQ772196; the bacterial strain was identified as Bacillus cereus DS1 (PQ772196). Phylogenetic tree was subsequently created utilizing MEGA 11 software to analyse its evolutionary relationships (Fig. 2).
Bacterial drought stress tolerance
Ethylene levels in plants are influenced by both biotic and abiotic stresses, which in turn play a key role in modulating various physiological processes,46 reducing root and shoot length under stress conditions. In ethylene biosynthesis, S- adenosylmethionine (S-AdoMet) is converted by 1-aminocyclopropane-1-carboxylate synthase (ACS) to 1- aminocyclopropane-1-carboxylate (ACC), which is the precursor of ethylene. The ACC is degraded by ACC deaminase, decreases ethylene-induced damage, ameliorates plant stress, and promotes plant growth.47
The bacteria was identified as Bacillus cereus (PQ772196) was able to produce ACC deaminase enzyme confirmed by plate assay, the blue color zone around the bacterial colony in the ACC minimal agar plate with pH indicator dye (bromothymol blue) indicative of ACC deaminase production. The isolated bacteria also effectively produce plant growth-promoting traits in normal and stress-mediated conditions.
It solubilised phosphate, with a solubilization index of 2.57 under non-stress conditions and 2.42 under stress conditions. The isolate also produced indole-3-acetic acid, ammonia, hydrogen cyanide, and siderophores, and it was capable of nitrogen fixation in both stress and non-stress environments (Table 2).
 |
Figure 2: Phylogenetic tree of Bacillus cereus DS1 PQ772196 |
Table 2: Plant growth-promoting factors producing ability of Bacillus cereus DS1 PQ772196 under stress and non-stress conditions
| Trait | Non-stress condition | Stress condition |
| Phosphate | + | + |
| Indole acetic acid | + | + |
| HCN | + | + |
| Ammonia | + | + |
| Nitrogen fixation ability | + | + |
| Siderophore | + | + |
| ACC deaminase | + | + |
“+” Positive, “-” Negative
Seed germination study
Drought stress hampers seed germination, although the severity and adverse effects differ across species.48 The PGPR enhances plant growth by producing different growth regulators, stimulating the growth directly, aiding in nodulation, or indirectly stimulating nodulation.49,50 However, PGPR solubilizes the minerals by producing organic acids, mineralizes and enhances nutrient uptake.51 In the study on seed germination under drought conditions, wheat seeds were bioprimed using a suspension of the isolated strain Bacillus cereus DS1, and the germination percentage was subsequently evaluated. When wheat seeds were bioprimed with Bacillus cereus DS1 in the absence of PEG 8000, the germination rate matched the control experiment, with 100% of the seeds successfully germinating. A 5% PEG 8000 solution, inducing drought stress at -0.47 MPa, was applied, and the seed germination rate was observed. Under this stress condition, germination occurred at a reduced rate of 14%. However, when the stress was combined with the bacterial suspension, germination significantly improved, reaching 88%. This evidence indicates that the bacteria enhance seed germination under drought conditions.
 |
Figure 3: wheat seeds were treated with A) 5% PEG 8000, B) Distilled water, C) Only bacterial suspension, D) 5% PEG 8000 in combination with bacteria. |
Table 3: Germination performance of seeds under control, bacterial, PEG, and combined treatments
| Sr. No. | Treatment | Replicate 1 | Replicate 2 | Replicate 3 | SD | Total seeds germinated |
| 1 | Control | 14 | 14 | 14 | 0 | 42 |
| 2 | Bacterial suspension | 14 | 14 | 14 | 0 | 42 |
| 3 | 5% PEG | 2 | 3 | 1 | 0.82 | 6 |
| 4 | Bacteria + 5% PEG | 12 | 12 | 13 | 0.47 | 37 |
Germination in the control set was considered as 100% for comparison.
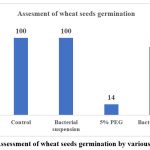 |
Figure 4: Assessment of wheat seeds germination by various treatment. |
Bacillus cereus, as reported by De Oliveira et al. (2024),52 has been effectively utilised to enhance popcorn plants’ growth under drought conditions. This bacterial strain promotes notable morphological changes and enhances several key growth parameters—such as elevated chlorophyll content, improved chlorophyll fluorescence and maximum fluorescence intensity, greater stem diameter, expanded leaf area, longer and denser roots, increased biomass accumulation, and more efficient water use. Additionally, multiple studies 28,53–55 highlight the Bacillus genus as a promising candidate among plant growth-promoting bacteria, offering considerable potential for improving plant resilience and productivity. Bacillus cereus is widely recognised as a pathogenic species; however, some isolated strains have been reported to be environmentally safe and able to produce some plant growth-promoting traits, including IAA and ACC deaminase.56
The bacteria identified as Bacillus cereus DS1 may have the potential to reduce the severity of drought stress, enhance nutrient uptake, and make plants more stress-tolerant under drought conditions. This study primarily examined seed germination. However, a comprehensive exploration of Bacillus cereus DS1 for plant growth and crop yield improvement is also necessary.
Conclusion
To address water scarcity and reduce dependence on harmful agrochemicals, researchers are investigating an eco-friendly approach to activate plants’ innate defence mechanisms against abiotic stresses. PGPR are essential for boosting the growth of plants and increasing productivity under drought conditions. The Bacillus cereus DS1 isolated in this study has the potential to produce various plant growth-promoting traits under stress and non-stress conditions. Bacillus cereus DS1 has the potential to stimulate nutrient assimilation under water-deficient environments, as evidenced by a seed germination study. The outcomes of the present research suggest that Bacillus cereus DS1 could be useful in developing bioinoculants to boost crop production in arid and semi-arid areas. Consequently, this bacterium could be a valuable resource for enhancing global food security.
Acknowledgment
Authors gratefully acknowledge the National Collection of Industrial Microorganisms, CSIR-National Chemical Laboratory, Pune (India) for 16S rDNA sequencing.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
All datasets generated or analyzed during this study are included in the manuscript.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Snehal Maruti Patil: Data Collection, Methodology, Analysis,
- Shubham Dattatray Jadhav: Data Collection, Analysis
- Shubham Chandrakant Kavade: Data Collection, Analysis, Writing
- Rameshwar Vishnu Darade: Writing– Review & Editing.
- Ajitkumar Lole: Resources, Review & Editing.
- Ajaykumar Gangadhar Jadhav: Visualisation, Supervision, Review & Editing.
- Gajanan Motiram Shinde: Visualisation, Supervision, Project Administration, Writing– Original Draft, Review & Editing.
References
- Mirzabaev A, Bezner Kerr R, Hasegawa T, et al. Severe climate change risks to food security and nutrition. Climate Risk Management. 2023;39:100473. doi:10.1016/j.crm.2022.100473
CrossRef - Austin RB. Prospects for improving crop production in stressful environments. In: Jones HG, Flowers TJ, Jones MB, eds. Plants under Stress. 1st ed. Cambridge University Press; 1989:235-248. doi:10.1017/CBO9780511661587.014
CrossRef - Vurukonda SSKP, Vardharajula S, Shrivastava M, SkZ A. Enhancement of drought stress tolerance in crops by plant growth promoting rhizobacteria. Microbiological Research. 2016;184:13-24. doi:10.1016/j.micres.2015.12.003
CrossRef - Hsiao, A. Effect of water deficit on morphological and physiological characterizes in Rice (Oryza sativa). J Agric. 2000;3:93-97.
- Rahdari P, Hoseini S M. Drought stress, a review. Int J Agron Plant Prod. 2012;3(10):443-446.
- Jaleel CA, Paramasivam Manivannan, Abdul Wahid, Muhammad Farooq, Hameed Jasim Al-Juburi, Ramamurthy Somasundaram and Rajaram Panneerselvam. Drought Stress in Plants: A Review on Morphological Characteristics and Pigments Composition. Int J Agric Biol. 2009;11(1):100-105.
- Barber SA. Soil Nutrient Bioavailability: A Mechanistic Approach. John Wiley & Sons; 1995.
- Selvakumar G, Panneerselvam P, Ganeshamurthy AN. Bacterial Mediated Alleviation of Abiotic Stress in Crops. In: Maheshwari DK, ed. Bacteria in Agrobiology: Stress Management. Springer Berlin Heidelberg; 2012:205-224. doi:10.1007/978-3-642-23465-1_10
CrossRef - Cruz De Carvalho MH. Drought stress and reactive oxygen species: production, scavenging and signaling. Plant Signaling & Behavior. 2008;3(3):156-165. doi:10.4161/psb.3.3.5536
CrossRef - Gigon A, Matos AR, Laffray D, Zuily-Fodil Y, Pham-Thi AT. Effect of Drought Stress on Lipid Metabolism in the Leaves of Arabidopsis thaliana (Ecotype Columbia). Annals of Botany. 2004;94(3):345-351. doi:10.1093/aob/mch150
CrossRef - Abid M, Ali S, Qi LK, et al. Physiological and biochemical changes during drought and recovery periods at tillering and jointing stages in wheat (Triticum aestivum L.). Sci Rep. 2018;8(1):4615. doi:10.1038/s41598-018-21441-7
CrossRef - Khan AA, Wang YF, Akbar R, Alhoqail WA. Mechanistic insights and future perspectives of drought stress management in staple crops. Front Plant Sci. 2025;16:1547452. doi:10.3389/fpls.2025.1547452
CrossRef - Zhou W, Li M, Achal V. A comprehensive review on environmental and human health impacts of chemical pesticide usage. Emerging Contaminants. 2025;11(1):100410. doi:10.1016/j.emcon.2024.100410
CrossRef - Ferreyra-Suarez D, García-Depraect O, Castro-Muñoz R. A review on fungal-based biopesticides and biofertilizers production. Ecotoxicology and Environmental Safety. 2024;283:116945. doi:10.1016/j.ecoenv.2024.116945
CrossRef - Naidu R, Biswas B, Willett IR, et al. Chemical pollution: A growing peril and potential catastrophic risk to humanity. Environment International. 2021;156:106616. doi:10.1016/j.envint.2021.106616
CrossRef - Ogidi OI, Izah SC, Akpan UM. Food Quality and Agrochemical Use: Integrated Monitoring, Assessment, and Management Policies. In: Ogwu MC, Chibueze Izah S, eds. One Health Implications of Agrochemicals and Their Sustainable Alternatives. Vol 34. Sustainable Development and Biodiversity. Springer Nature Singapore; 2023:411-440. doi:10.1007/978-981-99-3439-3_15
CrossRef - Hendry GAF. Oxygen, free radical processes and seed longevity. Seed Sci Res. 1993;3(3):141-153. doi:10.1017/S0960258500001720
CrossRef - Nair AS, Abraham TK, Jaya DS. Studies on the changes in lipid peroxidation and antioxidants in drought stress induced cowpea (Vigna unguiculata L.) varieties. J Environ Biol. 2008;29(5):689-691.
- Sgherri CLM, Maffei M, Navari-Izzo F. Antioxidative enzymes in wheat subjected to increasing water deficit and rewatering. Journal of Plant Physiology. 2000;157(3):273-279. doi:10.1016/S0176-1617(00)80048-6
CrossRef - Smirnoff N. The role of active oxygen in the response of plants to water deficit and desiccation. New Phytologist. 1993;125(1):27-58. doi:10.1111/j.1469-8137.1993.tb03863.x
CrossRef - Iqbal N, Masood A, Khan NA. Phytohormones in Salinity Tolerance: Ethylene and Gibberellins Cross Talk. In: Khan NA, Nazar R, Iqbal N, Anjum NA, eds. Phytohormones and Abiotic Stress Tolerance in Plants. Springer Berlin Heidelberg; 2012:77-98. doi:10.1007/978-3-642-25829-9_3
CrossRef - Nakashima K, Yamaguchi-Shinozaki K, Shinozaki K. The transcriptional regulatory network in the drought response and its crosstalk in abiotic stress responses including drought, cold, and heat. Front Plant Sci. 2014;5. doi:10.3389/fpls.2014.00170
CrossRef - Naylor D, Coleman-Derr D. Drought Stress and Root-Associated Bacterial Communities. Front Plant Sci. 2018;8:2223. doi:10.3389/fpls.2017.02223
CrossRef - Chandran H, Meena M, Swapnil P. Plant Growth-Promoting Rhizobacteria as a Green Alternative for Sustainable Agriculture. Sustainability. 2021;13(19):10986. doi:10.3390/su131910986
CrossRef - Basu A, Prasad P, Das SN, et al. Plant Growth Promoting Rhizobacteria (PGPR) as Green Bioinoculants: Recent Developments, Constraints, and Prospects. Sustainability. 2021;13(3):1140. doi:10.3390/su13031140
CrossRef - Farhat F, Tariq A, Waseem M, et al. Plant Growth Promoting Rhizobacteria (PGPR) Induced Improvements in the Growth, Photosynthesis, Antioxidants, and Nutrient Uptake of Rapeseed (Brassica napus L.). Gesunde Pflanzen. 2023;75(5):2075-2088. doi:10.1007/s10343-023-00845-0
CrossRef - Glick BR. The enhancement of plant growth by free-living bacteria. Can J Microbiol. 1995;41(2):109-117. doi:10.1139/m95-015
CrossRef - Sood G, Kaushal R, Chauhan A, Gupta S. Effect of conjoint application of indigenous PGPR and chemical fertilizers on productivity of maize ( Zea mays L.) under mid hills of Himachal Pradesh. Journal of Plant Nutrition. 2018;41(3):297-303. doi:10.1080/01904167.2017.1381116
CrossRef - Bano Q, Ilyas N, Bano A, Zafar N, Akram A, Hassan F. Effect of Azospirillum inoculation on maize Zea mays l under drought stress. Pak J Bot. 2013;45(1):13-20.
- Sandhya V, Ali SkZ, Grover M, Reddy G, Venkateswarlu B. Effect of plant growth promoting Pseudomonas spp. on compatible solutes, antioxidant status and plant growth of maize under drought stress. Plant Growth Regul. 2010;62(1):21-30. doi:10.1007/s10725-010-9479-4
CrossRef - Turan M, Güllüce M, Çakmak R, Şahin F. Effect of plant growth-promoting rhizobacteria strain on freezing injury and antioxidant enzyme activity of wheat and barley. Journal of Plant Nutrition. 2013;36(5):731-748. doi:10.1080/01904167.2012.754038
CrossRef - Marulanda A, Porcel R, Barea JM, Azcón R. Drought Tolerance and Antioxidant Activities in Lavender Plants Colonized by Native Drought-tolerant or Drought-sensitive Glomus Species. Microb Ecol. 2007;54(3):543-552. doi:10.1007/s00248-007-9237-y
CrossRef - Liu J, Shang W, Zhang X, Zhu Y, Yu K. Mn accumulation and tolerance in Celosia argentea Linn.: A new Mn-hyperaccumulating plant species. Journal of Hazardous Materials. 2014;267:136-141. doi:10.1016/j.jhazmat.2013.12.051
CrossRef - Odjegba VJ, Adeniyi AM. Responses of Celosia argentea L. to simulated drought and exogenous salicylic acid. Nat Sci. 2012;10(12):252-258.
- Soil Testing Methods Manual. FAO; 2020. doi:10.4060/ca2796en
CrossRef - Michel BE. Evaluation of the Water Potentials of Solutions of Polyethylene Glycol 8000 Both in the Absence and Presence of Other Solutes. Plant Physiol. 1983;72(1):66-70. doi:10.1104/pp.72.1.66
CrossRef - Patil C, Suryawanshi R, Koli S, Patil S. Improved method for effective screening of ACC (1-aminocyclopropane-1-carboxylate) deaminase producing microorganisms. Journal of Microbiological Methods. 2016;131:102-104. doi:10.1016/j.mimet.2016.10.009
CrossRef - Penrose DM, Glick BR. Methods for isolating and characterizing ACC deaminase‐containing plant growth‐promoting rhizobacteria. Physiologia Plantarum. 2003;118(1):10-15. doi:10.1034/j.1399-3054.2003.00086.x
CrossRef - Premono ME, Moawad A M, Vlek P L G. Effect of phosphate-solublizing Pseudomonas putida on the growth of maize and its survival in the rhizosphere. Indonesian Journal of Crop Science. 1997;11(1):13-23.
- Louden BC, Haarmann D, Lynne AM. Use of Blue Agar CAS Assay for Siderophore Detection. J Microbiol Biol Educ. 2011;12(1):51-53. doi:10.1128/jmbe.v12i1.249
CrossRef - Bakker AW, Schippers B. Microbial cyanide production in the rhizosphere in relation to potato yield reduction and Pseudomonas SPP-mediated plant growth-stimulation. Soil Biology and Biochemistry. 1987;19(4):451-457. doi:10.1016/0038-0717(87)90037-X
CrossRef - Lotfi N, Soleimani A, Çakmakçı R, Vahdati K, Mohammadi P. Characterization of plant growth-promoting rhizobacteria (PGPR) in Persian walnut associated with drought stress tolerance. Sci Rep. 2022;12(1):12725. doi:10.1038/s41598-022-16852-6
CrossRef - Dye DW. The inadequacy of the usual determinative tests for the identification of Xanthomonas spp. New Zealand Journal of Science. 1962;5(4):393-416.
- Dey R, Pal KK, Bhatt DM, Chauhan SM. Growth promotion and yield enhancement of peanut (Arachis hypogaea L.) by application of plant growth-promoting rhizobacteria. Microbiological Research. 2004;159(4):371-394. doi:10.1016/j.micres.2004.08.004
CrossRef - Fiodor A, Ajijah N, Dziewit L, Pranaw K. Biopriming of seed with plant growth-promoting bacteria for improved germination and seedling growth. Front Microbiol. 2023;14:1142966. doi:10.3389/fmicb.2023.1142966
CrossRef - Hardoim PR, Van Overbeek LS, Elsas JDV. Properties of bacterial endophytes and their proposed role in plant growth. Trends in Microbiology. 2008;16(10):463-471. doi:10.1016/j.tim.2008.07.008
CrossRef - Glick BR. Modulation of plant ethylene levels by the bacterial enzyme ACC deaminase. FEMS Microbiology Letters. 2005;251(1):1-7. doi:10.1016/j.femsle.2005.07.030
CrossRef - Li H, Li X, Zhang D, Liu H, Guan K. Effects of drought stress on the seed germination and early seedling growth of the endemic desert plant Eremosparton songoricum (Fabaceae). EXCLI J. 2013;12:89-101.
- Parewa HP, Yadav J, Rakshit A, Meena VS, Karthikeyan N. Plant growth promoting rhizobacteria enhance growth and nutrient uptake of crops. Agric Sustain Dev. 2014;2(2):101-116.
- Slimani A, Raklami A, Oufdou K, Meddich A. Isolation and Characterization of PGPR and Their Potenzial for Drought Alleviation in Barley Plants. Gesunde Pflanzen. 2023;75(2):377-391. doi:10.1007/s10343-022-00709-z
CrossRef - Tinker PB. The role of microorganisms in mediating and facilitating the uptake of plant nutrients from soil. In: Tinsley J, Darbyshire JF, eds. Biological Processes and Soil Fertility. Springer Netherlands; 1984:77-91. doi:10.1007/978-94-009-6101-2_7
CrossRef - De Oliveira UA, Amaral Junior ATD, Kamphorst SH, et al. Bacillus cereus: An Ally Against Drought in Popcorn Cultivation. Microorganisms. 2024;12(11):2351. doi:10.3390/microorganisms12112351
CrossRef - Azeem M, Javed S, Zahoor AF. Bacillus Species as Potential Plant Growth Promoting Rhizobacteria for Drought Stress Resilience. Russ J Plant Physiol. 2023;70(4):59. doi:10.1134/S1021443723600538
CrossRef - Lahlali R, Ezrari S, Radouane N, et al. Bacillus spp.-Mediated Drought Stress Tolerance in Plants: Current and Future Prospects. In: Islam MT, Rahman M, Pandey P, eds. Bacilli in Agrobiotechnology. Bacilli in Climate Resilient Agriculture and Bioprospecting. Springer International Publishing; 2022:487-518. doi:10.1007/978-3-030-85465-2_21
CrossRef - Md Gulzar AB, Mazumder PB. Seed priming and subsequent inoculation of drought-tolerant plant growth-promoting rhizobacteria Bacillus tropicus KH90 improves rice (Oryza sativa L.) plant health under drought and non-drought conditions. CEREAL RESEARCH COMMUNICATIONS. Published online August 2, 2024. doi:10.1007/s42976-024-00559-7
CrossRef - Kulkova I, Dobrzyński J, Kowalczyk P, Bełżecki G, Kramkowski K. Plant Growth Promotion Using Bacillus cereus. IJMS. 2023;24(11):9759. doi:10.3390/ijms24119759
CrossRef

