Introduction
Increasing unfavorable environmental conditions are hampering crop production globally. Salt stress is significant environmental factor that inhibit crop production worldwide. About 33% of the agricultural land is affected by soil salinity, and almost 52 million ha in South Asia is salt-affected.1 Saline soils are referred by high concentrations of Na+ ion with strongest anion of chloride consequently in electrical conductivity more than 4 dS m−1.2
Generally, salt stress declined plant growth, biomass accumulation and grain yield via osmotic, ionic and oxidative stress in plants.3-5 Salt stress boosted the Na+ ion in cells, reducing the normal function of K+ ion and impact on cellular metabolism.6 Homeostasis of Na+ and K+ ions and the Na+/K+ ratio are vital for salt tolerance in plant, and high Na+ ion accumulation interferes K+ homeostasis in plant cells by the ability of Na+ ion to compete for K+ binding sites.7 Moreover, plants have developed processes to uptake of osmolytes such as proline, which act as osmoprotectants and have a capable of mitigating osmotic stress through stabilization of proteins and shielding cellular functions by quenching reactive oxygen species (ROS).8 In higher plants, chlorophyll content normally decreases in salt-susceptible plants.9 Stomatal closure is one of the reactions when plants are exposed to salt stress, which leads to reduces of photosynthesis, biomass and seedling growth. Interference of photosynthetic processes increases the formation of ROS, leading to degradation of cell membrane.10 Moreover, plant growth is retarded because of lower relative water content (RWC), which caused by the reduction in cytoplasmic volume and the loss of cell turgor.11
Rice (Oryza sativa L.) is the key cereal crop that serves over half of the global population and is a moderately salt-sensitive crop.12,13 Hence, the current investigation was to evaluate the effect of salt stress on physiological and agronomical attributes of dissimilar rice varieties.
Materials and Methods
Growth Condition, Planting Materials, Treatments and Design of the Experiment
A pot experiment was carried out in Bangladesh Open University (90°38¢N, 23°95¢E). Two rice (Oryza Sativa L.) varieties BRRI dhan97 and BRRI dhan100 were used for the experiment. The BRRI dhan97 variety is a salt tolerant and BRRI dhan100 with salt-sensitive. Thirty six day old seedlings were transplanted into plastic pots (30.5 cm in diameter) with 12 kg of air-dried soil, with two seedlings per pot. The tested soil comprised with soil pH of 6.5, organic carbon (%) of 1.686, organic matter (%) of 2.908, total N (%) of 0.17, exchangeable K 0.27 meq/100 g soil, available P, S, and Zn respectively of 12.9, 25.01, and 9.07 ppm. Each pot received14 recommended fertilizer doses (Urea, TSP, MoP, Gypsum and ZnSO4 at the rates of 138, 51, 63, 60 and 4 kg ha-1, respectively). The study was conducted by following factorial Randomized Complete Block Design (RCBD) having three replications. The study comprised of following treatments – 0 mM NaCl and 80 mM NaCl. At panicle initiation stage, plants were irrigated with salt solution at each 2-day interval for 10 days. After 10 days, different physiological and agronomical parameters were measured.
Morphological trait
Determination of Biomass
Shoots were detached from root and the shoot dry weight (oven-dried at 70ºC for 72 h) were determined using a weighing scale.
Physiological and biochemical traits
Determination of Photosynthetic Pigments
Leaf (100 mg, fresh weight) was completely extracted with 10 ml of 80% acetone. The absorbance of the extract solution was monitored at 645, 663, and 470 nm, respectively, for chlorophyll a, chlorophyll b, and carotenoids content. The following equations were used for calculation.15
Chlorophyll a = 11.75 A663 2.350 A645,
Chlorophyll b = 18.61 A645 3.960 A663,
Total chlorophyll a+b = chlorophyll a + chlorophyll b and
Total carotenoid = (1000A470 2.270 Chl a 81.4 Chl b)/227
Determination of Relative Water Content (RWC)
Leaf RWC was measured by cutting the leaf; leaves were immediately weighed to obtain the fresh weight (FW) of leaf. The leaves were placed in 50 ml distilled water containing Petridis (110 mm) and kept in room temperature for 4 hours as described by Sairam.16 After 4 hours, the leaves were blotted with tissue paper and weighed to obtain the turgid weight (TW). Then the leaves were heated in oven at 70°C for 48hr to get dry weight (DW). The RWC was calculated using the following equation:
RWC (%) = [(FW-DW) / (TW-DW)] ×100
Estimation of Proline Content
Proline content was estimated according to the procedure of Bates.17 Fifty- milligram (50 mg) leaf samples were ground with sulfosalicylic acid (3 % w/v) by the mortar and it was centrifuged (12000 g for 15 min at 4°C). The sample was mixed with glacial acetic acid and ninhydrin reagent and incubated (95°C for 1 h). The reaction mixture was cooled in an ice. The solution was mixed with 4 ml toluene and then vortex. The chromophore absorbance was observed at 520 nm. A standard curve was prepared by L-proline.
Estimation of Na+ and K+ Content
Straw Na+ and K+ contents were estimated by a Na+ and K+ meter (Horiba, Kyoto, Japan). The straw was grinded with grinder and then straw sample was mixed with distilled water. The mixed sample was heated at 95°C for 30 minutes and cold at room temperature. The sample was put in sensor of the ion meter and data values were stated as ppm.
Measurement of Grain Weight
At maturity stage, panicle were collected to determine grain weight at 14% moisture.
Statistical Analysis
Two-way ANOVA with the statistix10 software (USA) was used for data analyze. The LSD test at P < 0.05 level of significance was used to compared the treatment means.
Results
Plant Biomass and Grain Weight of Rice Varieties under Salt Stress
The shoots of BRRI dhan100 showed significant reduction by 12.62% in dry weight at 80 mM concentration of NaCl, while BRRI dhan97 exhibited non-significant effect compared to control plants (Fig. 1A). The salinity stress reduced grain weight in both rice varieties, whereas more reduction was found in salt-sensitive rice variety (p < 0.05, Fig. 1B). Salt stress declined the grain weight of BRRI dhan97 rice variety by 70.46% and BRRI dhan100 rice variety by 84.49%.
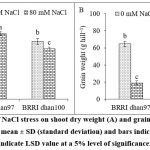 |
Figure 1: Effect of NaCl stress on shoot dry weight (A) and grain weight (B) of rice varieties. Data are mean ± SD (standard deviation) and bars indicate SD. Error bars indicate LSD value at a 5% level of significance.
|
Photosynthetic Pigments of Rice Varieties under Salt Stress
In this study, salt stress had a substantial effect on the photosynthetic pigments of rice varieties (p < 0.05, Figs. 2A-D). Compared with control, salt stress decreased Chl a, Chl b, Chl a+b and carotenoid of salt tolerant BRRI dhan97 by 14.02%, 22.95%, 16.25%% and 15%, respectively. In addition, under salt stress, the Chl a, Chl b, Chl a+b and carotenoid content of salt-sensitive BRRI dhan100 rice variety decreased by 68.57%, 67.79% %, 68.41% and 32.15%, respectively compared to control. The highest decrease was found by BRRI dhan100 than BRRI dhan97.
 |
Figure 2: Effect of NaCl stress on Chl a (A), Chl b (B), Chl a+b (C) and carotenoid content (D) of rice varieties. Data are mean ± SD (standard deviation) and bars indicate SD. Error bars indicate LSD value at a 5% level of significance. |
Relative Water Content and Osmoregulatory Substance of Rice Varieties under Salt Stress
Salt stress did not decrease RWC in salt-tolerant BRRI dhan97 rice variety. However, salt stress significantly declined the RWC of BRRI dhan100 by 27.50% compared to that of the control (Fig. 3A). Application of NaCl resulted in a significant effect on proline content of rice varieties (p < 0.05). Salt stress significantly increased proline content in BRRI dhan97 and BRRI dhan100, respectively, by 153.11% and 161.74% relative to control (Fig. 3B). However, proline content was more in salt-sensitive rice than in salt-tolerant rice.
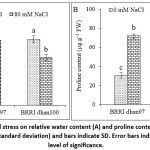 |
Figure 3: Effect of NaCl stress on relative water content (A) and proline content (B) of rice varieties. Data are mean ± SD (standard deviation) and bars indicate SD. Error bars indicate LSD value at a 5% level of significance. |
Na+, K+ and Na+/K+ ratio of Rice Varieties under Salt Stress
The content of Na+ ion in rice varieties was significantly affected under salt stress condition (Fig. 4A). Comparison with control, the Na+ content significantly increased by 2071% and 2241% in BRRI dhan97 and BRRI dhan100, respectively under salt stress. The content of K+ ion in rice varieties BRRI dhan97 and BRRI dhan100 was significantly decreased by 57.67% and 54.22%, respectively, compared to control plant under salt stress condition (Fig. 4B). The Na+/K+ ratio in straw was significantly variations by salt stress in both varieties. Compared to the control, the Na+/K+ ratio of BRRI dhan97 and BRRI dhan100 were increased by 4296.45% and 5910.27%, respectively, under salt stress. Overall, the Na+ and Na+/K+ ratio increased by 8.20% and 37.56% more in BRRI dhan100, respectively than BRRI dhan97 under salt stress (Figs. 4A and C).
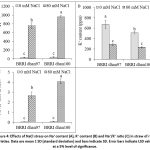 |
Figure 4: Effects of NaCl stress on Na+ content (A), K+ content (B) and Na+/K+ ratio (C) in straw of rice varieties. Data are mean ± SD (standard deviation) and bars indicate SD. Error bars indicate LSD value at a 5% level of significance. |
Discussion
Salinity stress affected the biomass production in both the rice cultivars. Results of the previous studies by Jahan18 and Xu19 reported that salt tolerant rice varieties, BINA dhan-8, and DJWJ, NSIC and JFX produced higher biomass than salt-sensitive rice varieties, BRRI dhan29, and XD2H, HKN and HHZ, respectively. Our results are in agreement with these findings and show that salt-tolerant BRRI dhan97 accumulate more biomass production than the salt-sensitive BRRI dhan100 under salt stress conditions. Overall, salt-sensitive rice variety showed 20.05% more grain weight decline than salt-tolerant rice variety under salt stress condition. This results consistent with the results of Xu19 who reported that salt-sensitive rice genotypes reduced more grain yield than salt-tolerant under salt stress.
Chlorophyll is a pigment crucial for light capture and photosynthesis.20 The degradation of photosynthetic pigments by salt stress was higher in BRRI dhan100 than the BRRI dhan97, suggesting the ability of this attribute to differentiate the response of salt-tolerant and salt-sensitive variety. These results are in agreement with the results of Monsur21 who found that rice plant treated with salt showed a significant decreased in Chl a, Chl b and carotenoid content in salt-sensitive rice than salt-tolerant rice. According to previous studies, salt-stressed salt-tolerant rice varieties were found to retention of chl content was greater than salt-sensitive varieties.22,23
Relative water content (RWC) was practiced in several investigations as major physiological parameters to assess salinity tolerance in rice.24-26 These results are accordance with the finding of Polash27 who reported that salinity reduces the relative water content in rice genotypes. The degradation of RWC was more in BRRI dhan29 than pokkali. Under salt stress condition, firstly plant face an osmotic stress that reduces water uptake from root to leaf. Moreover, ABA induced stomatal closure affects transpiration stream that direct low water uptake by roots, that cause low RWC in leaves.28 In this study, BRRI dhan97 probable facilitated the water uptake that performed more RWC comparison to the BRRI dhan100. These outcome suggested that BRRI dhan 97 rapidly changed osmotic adjustment to adapt osmotic stress and protect cellular water, while BRRI dhan100 showed a higher reduction in RWC due to its delayed response. An earlier investigation mentioned that salt-stressed salt-tolerant plants exhibited less decline in RWC than salt-sensitive plants.22 Similarly, in this study, the BRRI dhan97 performed higher salt tolerance than BRRI dhan100 by this parameter.
Proline accumulation in many plants under salt stress has been indicated to correlate with salt tolerance.29,30 The uptake of proline under salt stress differ among different rice varieties. In this study, proline content was boosted in salt-stressed rice seedlings compared to non-stressed plants which is higher noticeable in BRRI dhan100 than in BRRI dhan97, indicating that BRRI dhan100 plants was subjected to more acute osmotic stress than BRRI dhan97. Excess production and accumulation of proline in BRRI dhan100 is thought symptomatic of salt stress rather than a stress tolerance strategy. Similar results was found by Bhusan31 and Fang32 observed that salt-sensitive rice cultivar accumulated higher intracellular proline than salt-tolerant rice under salt stress conditions. In contrast Joseph33 observed that salt-tolerant rice varieties uptake more proline than salt-sensitive varieties. However, in this research, the uptake of proline is higher in salt-sensitive BRRI dhan100 (86.82µg g-1 FW) than salt-tolerant BRRI dhan97 (82.01µg g-1 FW) in saline condition, suggesting that salt-sensitive BRRI dhan100 needed to synthesize high levels of proline for osmotic adjustment under salt stress.
The sensitivity of rice to salinity is correlated with accumulation of Na+ to the root xylem via a apoplastic flow, leading to enough Na+ transport from roots to the shoot.34,35 Xu19 observed that salt-stressed salt-sensitive rice held up a high Na+/K+ ratio than salt-stressed salt-tolerant rice. However, salt-tolerant rice have higher Na+ exclusion ability than sensitive one via SOS1-dependent exclusion mechanism.36 Moreover, lesser Na+/K+ ratio in salt-tolerant rice, which can control Na+ sequestration in vacuoles and exclude Na+ ion via Na+/K+ antiporters.37 In addition, Zhang38 also observed that the salt-tolerant rice JYGY-1 has performed the minimum Na+/K+ ratio through exclusion of toxic apoplastic Na+ via salt overly sensitive 1 (OsSOS1) encoded Na+/K+ antiporter in plasma membrane.
Conclusion
Salt stress significantly affects photosynthetic pigments, Na+/K+ ratio and grain weight of both rice varieties and observed a more reduction in salt-sensitive rice variety. Consequently, biomass accumulation and relative water content significantly decreased in salt-stressed salt-sensitive rice. However, salt stress did not effect on biomass accumulation and RWC in salt-tolerant rice variety. Proline content increased significantly in both rice varieties, and the increase was greater in salt-stressed salt-sensitive rice. In conclusion, BRRI dhan97 was found to have strong salt tolerance than BRRI dhan100 in both agronomical and physiological traits.
Acknowledgement
The authors are thanks to BRRI authority for providing the seeds of rice varieties.
Funding Sources
The authors would like to admire to Bangladesh Open University for funding this research.
Conflicts of Interest
The authors have no conflict of interest about this manuscript.
Data Availability Statement
The manuscript incorporates all datasets examined throughout this study.
Ethical Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Author Contributions
Muhammad Abdus Sobahan: Conceptualization, Methodology, Writing – Original Draft.
Nasima Akter: Data Analysis, Writing – Review & Editing.
References
- Mandal S., Bhardwaj A., Ramadas S. Current status of research, technology response and policy needs of salt-affected soils in India-A review related papers identifying extension opportunities to enhance wheat productivity in north-east India Sendhil Ramadas. J Ind S Coastal Agr Res. 2018; 36:40-53.
- Ali S., Gautam R.K., Mahajan R., Krishnamurthy S.L., Sharma S.K., Singh R.K., Ismail A.M. Stress indices and selectable traits in SALTOL QTL introgressed rice genotypes for reproductive stage tolerance to sodicity and salinity stresses. Field Crops Res. 2013; 154: 65-73.
CrossRef - Yang Y., Guo Y. Elucidating the molecular mechanisms mediating plant salt-stress responses. New Phytol.2018; 217: 523-539.
CrossRef - Sikder R.K., Wang X., Jin D., Zhang H., Gui H., Dong Q., Pang N., Zhang X., Song M. Screening and evaluation of reliable traits of upland cotton (Gossypium hirsutumL.) genotypes for salt tolerance at the seedling growth stage. J. Cotton Res. 2020; 3:11.
CrossRef - Sadak M.S., Sekara A., Al-ashkar I., Habib-ur-Rahman M., Skalicky M., Brestic M., Kumar A., Sabagh A.E., Abdelhamid M.T. Exogenous aspartic acid alleviates salt stress induced decline in growth by enhancing antioxidants and compatible solutes while reducing reactive oxygen species in wheat. Front. Plant Sci.2022; 13: 987641.
CrossRef - Farooq M., Wahid A., Kobayashi N., Fujita D., Basra S.M.A. Plant drought stress: Effects, mechanisms and management. Agron Sustain Dev.2009; 29; 185-212.
CrossRef - Rejili M., Vadel A.M., Guetet A., Neffatti M. Effect of NaCl on the growth and the ionic balance K+/Na+ of two populations of Lotus creticus (L.) (Papilionaceae). S Afr J Bot. 2007; 73:623-631
CrossRef - Sasaki T., Matsumoto T., Yamamoto K., Sakata K., Baba T., Katayose Y., Wu J., Niimura Y., Cheng Z., Nagamura Y. The map-based sequence of the rice genome. Nature. 2005; 436: 793-800.
CrossRef - Heidari A., Bandehagh A., Toorchi M. (2014). Effects of NaCl stress on chlorophyll content and chlorophyll fluorescence in sunflower (Helianthus annuus L.) lines. Yüzüncü Yıl Univ J Agr Sci. 2014; 24:111-120.
CrossRef - Apel K., Hirt H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol. 2004; 55:373-399.
CrossRef - Vijayakumari K., Puthur J.T. γ-Aminobutyric acid (GABA) priming enhances the osmotic stress tolerance in Piper nigrum Linn. Plants subjected to PEG-induced stress. Plant G Reg. 2016; 78:57-67.
CrossRef - Shaheen R., Hood-Nowotny R.C. Carbon isotope discrimination: Potential for screening salinity tolerance in rice at the seedling stage using hydroponics. Plant Breed. 2005; 124: 220-224.
CrossRef - Joseph B., Jini D., Sujatha S. Biological and physiological perspectives of specificity in abiotic salt stress response from various rice plants. Asian J Agric Sci. 2010; 2 (3), 99-105.
CrossRef - BRRI (Bangladesh Rice Research Institute). Adhunik dhaner chash. Joydebpur, (Gazipur: Bangladesh Rice Research Institute) 2020; 1701. 106.
- Lichtenthaler H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Met Enzymol. 1987; 48: 350-382.
CrossRef - Sairam R.K., Rao K.V., Srivastava G.C. Differential response of wheat genotypes to long term salinity stress relation to oxidative stress, antioxidant activity and osmolyte concentration. Plant Sci. 2002; 163: 1037-1046.
CrossRef - Bates L.S., Waldren R.P., Teari D. Rapid determination of free proline for water stress studies. Plant Soil. 1973; 39: 205-207.
CrossRef - Jahan F., Bhusan D., Jahiruddin M., Murata Y., Hoque M.A. Improvement of salinity tolerance in rice during boro season by proline application. Prog Agr. 2018; 29: 295-303.
CrossRef - Xu Y., Bu W., Xu Y., Fei H., Zhu Y., Ahmed I., Nimir N., Zhou G., Zhu G. Effects of Salt Stress on Physiological and Agronomic Traits of Rice Genotypes with Contrasting Salt Tolerance. Plants. 2024; 13:1157.
CrossRef - Sawicki A., Willows R.D., Chen M. Spectral signatures of five hydroxymethyl chlorophyll a derivatives chemically derived from chlorophyll b or chlorophyll f. Photosynth. Res. 2019; 140: 115-127.
CrossRef - Monsur M.B., Ivy N.A., Haque M.M., Hasanuzzaman M., Sabagh A.E.L., Rohman M.M. Oxidative Stress Tolerance Mechanism in Rice under Salinity. Phyton-Int J Exp Bot. 2020; 89(3): 497-517.
CrossRef - Ma N.L., Lah W.A.C., Kadir N.A., Mustaqim M., Rahmat Z., Ahmad A., Lam S.D., Ismail M.R. Susceptibility and tolerance of rice crop to salt threat: Physiological and metabolic inspections. PLoS ONE. 2018; 13 (2):e0192732.
CrossRef - Sonone M., Mane A., Sawardekar S., Kunkerkar R. Consequences of salt stress on chlorophyll pigments of rice genotypes. The Pharm. Innov. J. 2023; 12: 3272-3275.
- Dionisio-Sese M.L., Tobita S. Antioxidant responses of rice seedlings to salinity stress. Plant Sci., 1998; 135:1-9.
CrossRef - Cha-um S., Trakulyingcharoen T., Smitamana P. Kirdmanee C. Salt Tolerance in two rice cultivars differing salt tolerant abilities in responses to iso-osmotic stress. Aust. J. Crop Sci. 2009; 3:221-230.
- Hoang, T.M.L., Williams, B., Khanna, H., Dale, J., Mundree, S.G. (2014). Physiological basis of salt stress tolerance in rice expressing the antiapoptotic gene SfIAP. Funct Plant Biol. 2014; 41:1168-1177.
CrossRef - Polash M.A.S., Sakil M.A., Tahjib-Ul-Arif M., Hossain M.A. Effect of salinity on osmolytes and relative water content of selected rice genotypes. Trop Plant Res. 2018; 5: 227-232.
CrossRef - Blatt M.R., Armstrong F. K+ channels for stomatal guard cells, abscisic acid evoked control of outward rectifier mediated by cytoplasmic pH. Planta. 1993; 191: 330-341.
CrossRef - Szabados L., Savoure A. Proline: A multifunctional amino acid. Trends Plant Sci., 2010; 15:89-97.
CrossRef - Kavi Kishor P.B., Sreenivasulu N. Is proline accumulation per se correlated with stress tolerance or is proline homeostasis a more critical issue? Plant Cell Environ. 2014; 37:300-311.
CrossRef - Bhusan D., Das D.K., Mahmud H., Yoshiyuki M., Hoque, M.A. Improvement of salt tolerance in rice (Oryza sativa L.) by increasing antioxidant defense systems using exogenous application of proline. Aust J Crop Sci. 2016; 10(1): 50-56.
- Fang X., Mo J., Zhou H. Shen X., Xie Y., Xu J., Yang S.Comparative transcriptome analysis of gene responses of salt-tolerant and salt-sensitive rice cultivars to salt stress. Sci Rep. 2023;13: 19065.
CrossRef - Joseph E.A., Radhakrishnan V.V., Mohanan K.V. A Study on the Accumulation of Proline- An Osmoprotectant Amino Acid under Salt Stress in Some Native Rice Cultivars of North Kerala, India. Uni J Agr Res. 2015; 3(1): 15-22.
CrossRef - Sobahan M.A., Arias C.R., Okuma E., Shimoishi Y., Nakamura Y., Hirai Y., Mori I. C., Murata Y. Exogenous proline and glycinebetaine suppress apoplastic flow to reduce Na+uptake in rice seedlings. Biosci Biotech Bioche.2009; 73(9): 2037-2042.
CrossRef - Faiyue B., Al-Azzawi M.J., Flowers T.J. A new screening technique for salinity resistance in rice (Oryza sativaL.) seedlings using bypass flow. Plant Cell Environ. 2012; 35:1099-1108.
CrossRef - Gerona M.E.B., Deocampo M.P., Egdane J.A., Ismail A.M., Dionisio-Sese M.L. Physiological responses of contrasting rice genotypes to salt stress at reproductive stage. Rice Sci. 2019; 26: 207-219.
CrossRef - Ismail A.M., Horie T. Genomics, physiology, and molecular breeding approaches for improving salt tolerance. Annu Rev Plant Biol. 2017; 68: 405-434.
CrossRef - Zhang Y., Fang J., Wu X., Dong L.Na+/K+Balance and Transport Regulatory Mechanisms in Weedy and Cultivated Rice (Oryza sativa L.) Under Salt Stress. BMC Plant Biol. 2018; 18: 375.
CrossRef
Abbreviations
|
FW TSP NaCl BRRI RCBD RWC ANOVA LSD ABA |
– – – – – – – – – |
Fresh Weight Triple Super Phosphate Muriate of Potash Sodium Chloride Bangladesh Rice Research Institute Randomized Complete Block Design Relative Water Content Analysis of Variance Least Significant Difference Abscisic Acid |

