Introduction
The extended use of chemical fertilizers and pesticides negatively impacts soil by decreasing water retention capacity, increasing salinity, and disrupting the balance of nutrients, ultimately affecting soil structure and fertility. Developing eco-friendly alternatives for harmful chemicals and pesticides to combat crop disease is one of the biggest challenges for microbiologists and plant pathologists. PGPR (plant growth-promoting rhizobacteria) occupies several ecological and practical niches within the soil rhizosphere and performs key functions in agroecosystems, such as producing phytohormones: indole acetic acid (IAA), GA3, and antimicrobial metabolites: lipopeptides, volatile organic compounds (VOCs), ammonia (NH3), hydrogen cyanide (HCN), and catalase, which enhance plant growth, inhibit phytopathogens, and strengthen the plant’s natural defences while performing soil bioremediation.1 The application of these beneficial microorganisms as biopesticides and biofertilizers is deemed the most beneficial method for more sensible and safer crop management to enhance soil fertility while effectively controlling plant pathogens.1-3 Several bacteria present in the rhizosphere have antagonistic properties against phytopathogens and are essential in managing plant health. These PGPRs utilize multiple mechanisms to enhance plant growth and increase resistance to various pathogens and abiotic stresses; thus, they serve as an alternative to harmful chemical pesticides for sustainable agriculture.4
Termite mounds are the nests of termites, made up of partly digested organic matter and their excrements. It is abundant in humic acids, which play a crucial role as terminal electron acceptors in respiration, fostering the growth of microbial life.5 Termites are important microbial agents that create substantial physicochemical changes in tropical and subtropical soils, and they are known as “ecosystem engineers.”6 Termite mounds are constructed from the feces of termites and soil, both of which are rich in minerals and organic matter.7 The rich nutrient content and presence of beneficial soil organisms in the soil of termite mounds have been demonstrated to improve crop yields, making it suitable for use as biofertilizers.2,8,9
Bacillus species offer numerous advantages over other organisms due to their ability to form endospores, allowing them to withstand harsh environmental conditions such as extreme acidity (pH), temperature, and osmotic stress. These traits make them excellent candidates for developing effective biopesticides. Bacillus spp. colonizes root surfaces, promotes plant growth, and causes the breakdown of fungal mycelia, making it a highly promising and safe biological agent.10 Alternaria alternata is a fungal pathogen that causes leaf spots and other fungal diseases in more than 380 plant species. It acts as an opportunistic pathogen on various hosts, leading to leaf spots, rots, and blights on different plant parts. It is distributed over a wide range of climatic conditions, so it can be found in many regions of the world. A. alternata infection disrupts the plant hormonal balance, notably causing a significant reduction in the concentrations of gibberellins, IAA, abscisic acid, and cytokinins11 A. alternata produces metabolites that inhibit seed germination and cause chlorosis, necrosis, discoloration, and wilting of seedlings.12 Alternaria pathogen diseases on plants cause yield losses, reduced quality of the crop, and are very difficult to control.13
Though there are studies on soil-based bacterial isolation and its antagonistic activities, there is no reported study based on termitarium soil-inhabited bacterial isolation and identification for its antifungal properties against the phytopathogenic A. alternata, and it is crucial to develop potent biocontrol inoculums that act as an antifungal agents and help promote plant growth without lowering crop productivity, therefore the key objective of this research is to investigate Bacillus strains from termite mounds for their potential to control the phytopathogen A. alternata and promote mung bean (Vigna radiata L) growth in vitro.
Materials and Methods
Fungal phytopathogen
Standard culture of the fungal pathogen A. alternata (ITCC-6778), responsible for leaf spot disease in multiple host plants, was obtained from the Indian Type Culture Collection (ITCC-IARI), Indian Agricultural Research Institute, New Delhi, India, and maintained in PDA medium at 4°C.
Soil Sampling and Bio-agent Isolation
The soil samples for this study were obtained from the termite mounds of Papum Pare district, Arunachal Pradesh, India. A total of 100 g of termitarium soil was collected in sterile polybags from each location. The collected soil sample was maintained at 4°C for future enumeration. To isolate endospore-forming Bacillus spp. soil samples were screened to eliminate debris, and 10g of soil was weighed and subjected to heat treatment at 80°C for 10 minutes. To prepare soil suspensions, the treated soil was infused with 95 mL of sterilized distilled water, followed by serial dilutions ranging from 10⁻¹ to 10⁻⁹. Using the spread plate technique, 100 µl of suspension was evenly spread plated in Nutrient Agar (NA) plates and incubated at 30°C for 24 hours. Post incubation, bacterial colonies were counted, and pure cultures were obtained through sub-culturing on NA plates. The isolated bacterial strains were preserved in duplicate on NA slants at 4°C for further tests.14
Antifungal Screening
The dual culture method evaluated all isolated bacterial strains for antifungal activity. A 0.5 cm diameter fungal disc of A. alternata (72hours-old culture) was placed at the center of a 9 cm PDA (potato dextrose agar) plate. The dish was divided into four quadrants, and each bacterial isolate was spot-inoculated in a quadrant 2.5 cm from the fungal disc using a sterile stick and incubated in an inverted position for 72 hours at 28±2°C and monitored every 12 hours. Strains that inhibited fungal growth were selected and preserved 19
In vitro Dual culture assay
The dual culture was performed to evaluate the in vitro antagonistic effect of the termitarium soil-inhabiting bacteria against the phytopathogen A. alternata. A 5 mm fungal disc was placed at the centre of the potato dextrose agar (PDA) plate, and a fresh bacterial culture was streaked 2.5 cm on both sides of the fungal disc. The plates were inverted and incubated at 28°C for five days. Measurements of the fungal growth were taken at 24 hours intervals. The radial inhibition was calculated as: PI = (R1 – R2) / R1 × 100. In this equation, PI= percentage inhibition; R1 = fungal radial growth on the control plate; R2 = fungal radial growth on the treated plate.20
Cell-Free Culture Filtrate (CFCF) tests
The antagonistic properties of the CFCF derived from bacterial culture were evaluated by agar well diffusion.21 Antifungal extracellular filtrates were collected from bacterial samples during their logarithmic growth phase. The bacteria were cultured in potato dextrose broth (PDB) and incubated in a shaker incubator at 175 rpm and 28°C for 72 hours. After incubation, the culture broth was centrifuged at 10000 rpm for 10 minutes, and the resulting supernatant was filtered through a 0.22 µm syringe filter to obtain cell-free culture filtrate (CFCF). A fungal disc (5 mm) was placed in the centre of each PDA plate, and 200 µl of the CFCF was added to wells (5 mm) located 2.5 cm away from the fungal disc, and incubated at 28°C for 48 to 72 hours. The measurement of the inhibition zone was taken, and the percentage inhibition was calculated as: Inhibition % (PI) = [(Fungal diameter in control – Fungal diameter in treatment) / Fungal diameter control] × 100, where PI = percentage of inhibition.
Morphological and Biochemical Analysis
Standard procedures were followed for the morphological and biochemical characterisation of the bacterial isolates.15, 16 For morphological analysis, individual bacterial colonies were examined.
Amylase production test
The test isolates were inoculated by streaking fresh culture onto starch agar plates with a sterile loop and then incubated at 37°C for 48 hours. After incubation, the plates were soaked with an iodine solution for 30 seconds. The excess iodine solution was poured out, and clear zones around the colonies were observed to indicate amylase production; a clear zone implies starch hydrolysis, whereas a blue-black zone indicates unhydrolyzed starch and the absence of enzyme activity.15
Catalase test
The test is conducted to determine whether the test organism produces catalase.74, 75 Freshly obtained bacterial isolates were inoculated into trypticase soy agar slants, with one slant left uninoculated as a control, and incubated at 35°C for 48 hours. After incubation, 3-4 drops of hydrogen peroxide were dropped into the slant at an angle and allowed to flow over the bacterial colony. The development of bubbles following the addition of hydrogen peroxide confirmed a catalase-positive reaction.15
Cellulase production test
The cellulase test screens the bacteria that can produce cellulase enzyme, which breaks down cellulose, a major component of the plant cell wall. Czapek-Mineral Salt agar medium was prepared with 0.5 % CMC. The bacterial isolates were inoculated using a sterile loop and incubated for 48 hours at 30°C. After the incubation period, the plates were treated with a 1% hexadecyl trimethyl ammonium bromide solution and examined for a zone around the bacterial colony. A clear zone indicates a catalase-positive reaction.15
Casein Hydrolysis
The Casein Hydrolysis Test is a biochemical test that determines bacteria’s capacity to generate caseinase enzymes. The bacterial isolates were inoculated in skim milk agar in a single streak of line, with an uninoculated control plate, and incubated upside down at 37°C for 48 hours. After incubation, observation of a clear zone around the bacterial colony indicates a positive casein hydrolysis reaction.15
Urease test
The urease test helps in the identification of microorganisms that can produce the urease enzyme.74,75 A 24-hour bacterial isolate was inoculated into a sterile urea agar slant. The inoculated urea slant was incubated for 48 hours at 37°C. The change in colour of the urea agar from pale yellow to pink/red/cerise indicated a positive reaction for the degradation of urease.15
IMViC Test
The IMViC tests consist of indole production, methyl red, Voges-Proskauer, and citrate utilization tests. 15 The indole test was conducted by inoculating the bacterial isolate in tryptone broth and incubating it at 30-37°C for 48 hours. After the incubation period, a few drops of Kovac’s reagent were added to the broth. The appearance of a red ring at the top of the broth indicates a positive result for indole production.
To perform the methyl red test, 5 mL of MR-VP broth was inoculated with the bacterial isolates and incubated at 35°C for 48 hours. After the incubation period, five drops of methyl red indicator were introduced into each tube. The Voges-Proskauer test is conducted using the same MR-VP broth that is employed in the methyl red test, and is incubated for 48 hours. After incubation, 12 drops of V-P reagent I and 3 drops of V-P reagent II were added to the other set of tubes. The mixture is shaken and allowed to stand for 30 minutes. In the MR test positive result is indicated by a colour change to red, and yellow coloration is a negative reaction, while crimson-to-ruby pink (red) colour indicates a positive VP reaction, and no colour change signifies a negative reaction.
Citrate test
The test assesses the capacity of bacteria to utilize citrate.74,75 The citrate utilization test was performed on Simmons citrate agar slant, where sodium citrate served as the only carbon source and bromothymol blue was used as the pH indicator. The bacterial isolate was inoculated using the stab-and-streak method, while an uninoculated slant was maintained as a control. A change in colour to blue from green, along with the growth of the bacterial isolate, indicated a positive result for citrate utilization, whereas no colour change indicated a negative result.15
Ammonia test
The ammonia test determines if bacteria can produce ammonia as a by-product of their metabolism, typically from the breakdown of protein or amino acids. To test for ammonia, 10 mL of peptone water (10 gl-1 peptone; 5 g l-1 sodium chloride) was taken in test tubes and inoculated with test isolates, the uninoculated tube was taken as the control and incubated at 36°C for 72 hours. After the incubation period, 0.5 mL of Nessler’s reagent was added to each culture tube. The development of a brown to yellow colour indicated the production of ammonia by the bacterial isolates.17
Hydrogen Cyanide (HCN) test
HCN test detects the ability of bacteria to produce hydrogen cyanide, a toxic secondary metabolite to inhibits the growth of plant pathogens or solubilizes phosphates, making them available to plants. King’s B agar enriched with 4.4 g of glycine was streaked with the test isolate. A sterile blotting paper soaked in picric acid (0.5%) and sodium carbonate (1%) was placed inside the lid of each plate, which was then sealed with parafilm. An uninoculated plate was included as a control. The plates were then incubated at 28°C for 24 to 48 hours. The appearance of brown colour on the filter paper, transitioning from yellow, was observed as evidence of HCN production.18
IAA tests
The indole test screens for the capacity of an organism to break down the amino acid tryptophan and create indole. It is employed as part of the IMViC procedures, a battery of tests aimed to discriminate among members of the family Enterobacteriaceae.74 For qualitative analysis of IAA production, 2 ml of the culture broth was centrifuged at 10000 rpm for 10 minutes. Following centrifugation, 1 mL of the resulting supernatant was carefully transferred into a separate test tube. In this test tube, 1 drop of ortho-phosphoric acid and 2 mL of Salkowski reagent (2 mL .5 M FeCl3, 49 mL 70% perchloric acid, and 49 mL distilled water) were added. it was then incubated at room temperature for 30 minutes in the dark. After the incubation, the development of pink coloration of the solution was indicative of positive for the positive synthesis of IAA.22- 24
In vitro PGPR Test
The in vitro plant growth promotion test was performed using mung bean (Vigna radiata L.) seeds obtained from the local market of Nirjuli, Arunachal Pradesh, India. The mung beans were first washed in flowing tap water, rinsed with distilled water, and left to be soaked in distilled water overnight. The seeds that had been soaked overnight were subjected to surface sterilization using a 2% sodium hypochlorite solution for five minutes. Following the sterilization treatment, the seeds were rinsed off with sterilized distilled water 2 to 3 times. The bacterial inoculum is prepared by culturing the isolates in LB broth in a shaker incubator at 170 rpm and 28°C for 24 hours. The sterilized seeds were immersed in the culture broth for 30 minutes. After treatment, the seeds were planted on agar plates, while control plates contained untreated seeds soaked in water and incubated at 28±2°C. Post-incubation, the shoot and root lengths were measured, recording both in centimetres from the root tip to the top of the stem.25
Results
Termite Mound Soil Sample Collection
The termite mounts soil samples were collected from five different locations: Midpu; Karsingsa; forest nursery (Department of Forestry, NERIST); IG Park; and Ganga Lake of Paum Pare district, Arunachal Pradesh, India (93°12′ E to 94°13′ E and 26°56′ N to 27°35′ N) (Fig. 1). 100 g each of the Termite mount soil samples were collected in a sterile polybag and stored at 4°C for the isolation of bacteria.
 |
Figure 1: Map of Study area: Papum Pare District, Arunachal Pradesh, India. (Sampling points are indicated on the map with a star). |
Isolation of Bioagents
A total of 175 endospore-forming bacilli strains were isolated from the five termite mount samples. Out of these, 42 isolates, showing differences based on the assessment of their morphological characteristics, were selected for further studies (Table 1; Fig. 2). The microbial count in termite mount samples varied to a great extent with locations. Among various sites, Karsingsa had the highest count (5.5×105 cfu g-1), whereas the Nursery, Department of Forestry had the lowest count (2.5×105 cfu g-1) on the Nutrient Agar (NA) medium.
Table 1: Enumeration of the bacterial population from the termitarium mount
| Sample No. | Collection site | ||
| Colony count (×105 cfu g-1) |
Colonies purified | ||
| TH1 | Midpu (Forest) | 3.3 | 07 |
| TH2 | Ganga Lake, Itanagar (Bamboo grove) | 2.9 | 04 |
| TH4 | Nursery, NERIST Nirjuli | 2.5 | 06 |
| TH5 | IG Park, Itanagar | 3.3 | 11 |
| TH6 | Karsingsa (Forest) | 5.5 | 14 |
| Total | 42 | ||
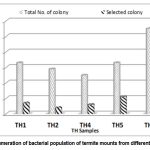 |
Figure 2: Enumeration of bacterial population of termite mounts from different soil samples. |
Screening of isolated bacteria for antifungal activity against A. alternata
A total of 42 bacterial isolates, selected based on their morphological characteristics, were screened for antifungal activity against the phytopathogen A. alternata. Out of these, eight isolates (TH1-C1, TH2-2, TH5-A, TH5-H, TH5-J, TH6-7, TH6-10, and TH6-15) exhibited antagonistic properties, forming clear inhibition zones against the phytopathogen. Specifically, one isolate, each from groups TH1 and TH2, and three from both groups TH5 and TH6, demonstrated antifungal activity. The remaining 34 isolates showed no antagonistic properties and were therefore excluded from further study. The six promising isolates were selected for identification and subsequent experimental analysis.
In vitro Dual Culture Assay
The effectiveness of the bacterial isolates in controlling A. alternata was evaluated by dual culture assay. The efficiency with which the bacterial isolate inhibits the radial growth of the phytopathogen in vitro is expressed as PI (percentage inhibition). Isolate TH5/J exhibited the highest average percentage of inhibition, followed by TH5/A, TH5/H, and TH1/C1, with values of 35.06%, 33.33%, 32.76%, and 29.88%, respectively (Fig. 3).
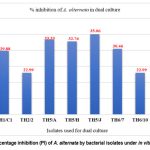 |
Figure 3: Percentage inhibition (PI) of A. alternata by bacterial isolates under in vitro conditions. |
CFCF test
CFCF (Cell Free Culture Filtrates) also inhibited A. alternata in vitro, but it showed weak antifungal activity. CFCF exhibited a limited inhibition of fungal growth in amended medium and in well diffusion techniques in vitro.
Morphological and Biochemical Characterization
Gram-staining and biochemical tests were conducted on six out of the eight tested bacterial isolates (TH1/C1, TH2/2, TH5/J, TH6/10, and TH6/15). All isolates were confirmed to be gram-positive. Through morphological assessment, Gram staining, and biochemical testing, these six isolates were identified as members of the genus Bacillus (Table 2).
Table 2: Morphological and biochemical tests for the selected isolates
| Bacterial isolates | ||||||
| Test | TH1/C1 | TH2/2 | TH5/J | TH5/A | TH6/10 | TH6/15 |
| Morphology | White, irregular, undulate | White, irregular, undulate | White, irregular, undulate | White, irregular, undulate | White, irregular, undulate | White, irregular, undulate |
| Cell Shape | Rod shaped | Rod shaped | Rod shaped | Rod shaped | Rod shaped | Rod shaped |
| Ammonia | + | + | − | − | − | + |
| HCN | + | + | − | − | + | + |
| Gram staining | + | + | + | + | + | + |
| Spore Staining | + | − | + | − | − | − |
| Amylase | + | + | + | + | + | + |
| Catalase | + | + | + | + | + | + |
| Cellulase | + | + | + | − | + | − |
| Casein | + | + | + | + | − | − |
| Urease | + | + | + | + | + | − |
| Indole | + | + | + | + | + | + |
| MR | + | + | + | + | + | + |
| VP | − | − | − | − | − | − |
| Citrate | − | − | − | − | − | − |
+ Positive result for the test; – Negative results for the test
IAA test
A qualitative study of IAA production showed that all the tested isolates synthesized IAA in LB broth culture medium. TH5/J produced the highest IAA as observed by the deep pink colouration, which supports the PGP in plants.
Plant Growth Promotion by Isolated Bacillus spp.
The influence of different bacteria on the promotion of Vigna radiata L. plant growth was assessed by measuring the height of the plants under various treatments of seeds in vitro. The untreated control group exhibited a growth height of 6.9 cm. In contrast, all treated groups showed a significant increase in growth. Among the treatments, TH2/2 exhibited the highest increase in plant growth with a total length of 17.4 cm, closely followed by the TH5/J and TH5/2 with a total height of 17.4 cm and 14.9 cm, respectively. The highest growth was exhibited in TH2/2, and the minimum plant growth promotion was observed in TH6/7 (Fig. 4).
Overall, the bacterial isolates significantly enhanced the growth of mung bean (V. radiata L.) plants compared to the control, indicating their potential as plant growth-promoting agents. The results suggest that certain isolates, particularly TH2/2 and TH5/J, are particularly effective in promoting growth in fenugreek. Further investigation is warranted to understand the mechanisms behind these growth-promoting effects.
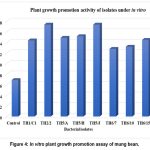 |
Figure 4: In vitro plant growth promotion assay of mung bean |
Discussion
Termite mounds are common in tropical agricultural landscapes but have received little attention for their influence on crop production. Their soil texture varies from top to bottom and differs from the surrounding soil. Termite mounds generally have lower organic matter and nitrogen but higher levels of phosphorus, calcium, potassium, magnesium, and sodium. Termites, key biological agents, modify soil properties through their mound-building activities, using salivary secretions to bind mineral particles.6, 26 Their activity also increases organic matter and alters clay composition in soils used for nest construction.27 Research highlights termites’ role in nutrient cycling and soil metabolism 28, 29, 30
Termites, which decompose surface litter, dry leaves, grass, lichens, and dung, contribute to 28% of earth’s annual net primary productivity.31 Their symbiotic relationships with microorganisms aid in nutrient recycling and influence soil properties in tropical ecosystems.32, 33 This contributes to a higher density and a greater diversity of microorganisms in termite mounds.9, 34 Termite mounds are built from feces and soil, are rich in nutrients, and have higher microbial density and diversity compared to the surrounding soil.34 Termite mound soil harbours a diverse range of beneficial bacteria, including Firmicutes, Proteobacteria, and Actinobacteria, promoting the activity of nitrogen fixers, decomposers, and sulfur-oxidizing microbes.35, 36 This soil, enriched with nutrients, enhances crop yield and can serve as a biofertilizer.9
Alternaria alternata is a fungus that is known to cause fungal blights, leaf spots, and rots in over 380 species of plants. It primarily affects stems, seeds, and seedlings and spreads through airborne spores. On tomatoes, symptoms include yellowing, browning, and defoliation, leading to significant crop losses, typically around 20%, but up to 70-80% in severe cases or combined with other diseases 37, 38. While fungicides are widely used to control Alternaria blights, resistance and environmental contamination remain challenges.39, 40 Research continues to optimize fungicide use and reduce spraying.41
In this study, 175 endospore-forming Bacillus strains were isolated from termite mounts in the Papum Pare district of Arunachal Pradesh, of which 8 isolates exhibited antifungal activity against the phytopathogenic A. alternata on initial screening. Through morphological and biochemical assessments, they were classified as belonging to the genus Bacillus. The findings of this study are in line with the previously reported findings, where 160 strains of Bacillus were isolated from termite mound soil.42 Termites compact the soil in the termite nest, thus limiting the soil organic matter availability to the microbes, resulting in a reduction of microbial population.27
Eco-friendly methods using biocontrol and resistance-inducing agents offer an effective alternative for plant disease suppression.13 Biological control through antagonistic microorganisms, like bacteria, is a promising strategy, providing eco-friendly benefits in disease control.43 Rhizobacteria, in particular, have revolutionized the biological management of plant pathogens by boosting systemic resistance and chemical defences against fungal infections.44, 45
Bacillus is ubiquitous, and commonly known for promoting plant growth and producing antibiotics, making it a key biocontrol agent.46, 47 Bacillus spp. offer advantages such as endospore formation and tolerance to extreme conditions, making them ideal candidates for biopesticides.48 They colonize plant roots, enhance growth, and lyse fungal mycelia, positioning them as safe and highly effective biocontrol agents.10 The production of antimicrobial lipopeptides by Bacillus spp., including surfactin, iturin, and fengycin, helps to inhibit phytopathogens, facilitate root colonization, and stimulate the host’s resistance.49, 50 Plants release root exudates that act as chemical signals, attracting Bacillus spp., which form biofilms on the roots, promoting plant health.51 The complex colonization process, driven by lipopeptides, plays a crucial role in biocontrol. 52
The isolates TH1/C1, TH2/2, TH5/A, TH5/H, TH5/J, TH6/7, TH6/10, and TH6/15 exhibited antagonism against the phytopathogen A. alternata, forming clear inhibition zones in dual culture. Isolate TH5/J exhibited the highest percentage inhibition at 35.06% of A. alternata, followed by TH5/A (33.33%) and TH5/H (32.76%). Numerous studies have demonstrated that Bacillus species isolated from various environments can inhibit the radial growth of A. alternata.53-55 Biological control is recognized as an appealing complement to existing disease management practices; however, the various aspects of this have not been thoroughly investigated.56 Bacillus spp. is a prominent biocontrol agent, due to its ability to produce spores, enabling it to thrive in stressful ecological conditions and combat phytopathogens in various situations. This characteristic ability provides a significant advantage in managing soil-borne fungal pathogens over extended periods or in unfavourable environments. In this context, Bacillus spp. can effectively control fungal phytopathogens, which form sclerotia that can remain dormant for many years.57 Bacillus strains effectively decrease the growth of the pathogenic fungus Fusarium solani,42 similarly, Bacillus isolate MSUA3 exhibited 78.9% and 72.8% growth inhibition of Fusarium oxysporum and Rhizoctonia solani, respectively.58 B. subtilis produces enzymes that break down the cell walls, such as protease, chitinase, and β-1,3-glucanase, which effectively antagonized the phytopathogenic strains of A. alternata.54
Sustainable agriculture is critical to meet the growing food and medical needs of the population. Traditional methods fall short, and modern farming often relies on synthetic chemicals that harm the environment and human health. Annually, plant diseases reduce global food production by 10-20%, leading to significant economic losses.59 PGPR plays a crucial role in sustainable farming by enhancing plant growth and offering protection against phytopathogens. These beneficial bacteria colonize plant roots, improving nutrient uptake and minimizing the impact of soil-borne diseases.60 PGPRs promote plant growth by performing several activities. One of their key roles is producing phytohormones such as IAA, cytokinins, and gibberellins, which stimulate root development and overall plant growth.61, 62 Additionally, some PGPRs enhance nitrogen fixation, providing plants with essential nitrogen for their growth.63 They also help in phosphate solubilization, converting inorganic phosphate into more accessible forms to plants and improving nutrient availability.64 Furthermore, PGPRs produce siderophores, which bind to iron, making them unavailable to harmful pathogens while simultaneously assisting plant iron uptake.65 They also release antimicrobial compounds such as antibiotics and enzymes that inhibit phytopathogens, offering natural disease protection to plants.66 Another important function of PGPR is induced systemic resistance (ISR), which stimulates the plant’s natural defences, making it more resistant to future pathogen attacks.67 In addition to these growth-promoting activities, PGPRs assist in detoxifying heavy metals, degrading pesticides, and supporting plant growth under stress conditions like salinity. By performing these functions, PGPRs reduce the need for chemical inputs, contributing to environmentally sustainable agriculture. The qualitative study of PGP activity of the isolated termite mount-inhabiting bacteria exhibited that they had a potential for PGP and as biofertilizers, as all the isolates tested positive for the synthesis of IAA in LB broth, with TH5/J producing the highest IAA. Of the 6 tested, three isolates: TH1/C1, TH2/2, and TH6/15 tested positive for ammonia. The production of IAA, one of the most quantitatively significant phytohormones, is a hallmark of PGPR.68 Bacillus pumilus MSUA3 produced IAA through the appearance of pink colour in filter paper bioassay with and without L-tryptophan.58 Plants provide PGPR with tryptophan to produce IAA, while Bacillus species generate significant amounts of IAA, which in turn aids in plant growth.69 B. subtilis isolated from the cardamom rhizosphere produced 43.25 µg mL of IAA when supplemented with L-tryptophan.70 The in vitro test for assessing PGP activity revealed that all bacterial isolates improved the growth of treated plants relative to the untreated control, with isolate TH2/2 exhibiting the highest increment, while the least increment was observed in isolate TH6/7 treated plants. In a greenhouse pot culture, the culture filtrate of Bacillus subtilis LY-1 significantly enhanced peanut growth, resulting in an increase of 30.77% in fresh mass and 27.27% in dry mass of the plants.71 Bacterization of turmeric rhizomes with Bacillus spp. resulted in increased fresh and dry weight of the turmeric rhizomes.42 The application of IAA-producing B. cereus UPMLH1 significantly boosted the number and length of shallot adventitious roots and enhanced shoot growth by 19% to 54%, in comparison, inoculation with the non-IAA-producing B. cereus UPMLH24 showed no significant impact.72 The application of PGPR to plants led to a marked improvement in shoot and root length, along with increased biomass for both shoots and roots. Furthermore, nitrogen content in shoots increased significantly by up to 76%, while root nitrogen content rose by up to 32% compared to the control group without inoculation. These PGPRs can be employed for the production of inoculants or biofertilizers to boost the growth and nutrient profiles of wheat and other crops in real-world agricultural scenarios. 73
Conclusion
The findings of this study indicate that the Bacillus spp. associated with the termite mount has the potential as a biocontrol agent for managing the leaf spot disease caused by A. alternata, while promoting plant growth. In addition to producing significant amounts of IAA, ammonia, and HCN that supported plant health, TH5/J showed the highest inhibition of A. alternata among the 42 isolated strains. Furthermore, TH2/2 and TH5/J markedly improved the in vitro development of mung beans. These results demonstrate the potential of Bacillus species generated from termite mounds as environmentally benign biopesticides and biofertilizers, providing a sustainable substitute for synthetic agrochemicals for increased soil health and crop output.
Further investigations are necessary to assess the efficiency of these bacterial isolates across multiple species of plants and geographical areas to enhance our understanding of their interactions with phytopathogens, plants, and soil characteristics.
Acknowledgment
All authors would like to thank the higher authorities of their respective departments, institutes, and universities for their cooperation and support during their studies. Authors ZK would like to acknowledge the RGNF-UGC fellowship for their support during the research.
Funding Sources
Author PK would like to thank Research and Development, Higher Education Department, U.P. Lucknow (letter no. 79/2024/1041/sattar-4-2024-001-4(33)/2023 dated 25-09-2024) for the financial support.
Conflicts of Interests
The authors declare no conflict of interest.
Data Availability Statement
No data was used for the research described in the article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Author Contributions
Pradeep Kumar, and Madhu Kaml.: Conceptualization & supervision, revision and editing of the manuscript.
Zeiwang Konyak: Performed experiments; original draft preparation.
Pradeep Kumar. funding acquisitions.
All authors have read and agreed to the published version of the manuscript.
References
- Maheshwari DK (Ed). Plant growth and health promoting bacteria. Springer Science & Business Media. 2010; 18
CrossRef - Enagbonma BJ, & Babalola OO. Potentials of termite mound soil bacteria in ecosystem engineering for sustainable agriculture. Annals of Microbiology. 2019; 69: 211-219.
CrossRef - Hasan A, Tabassum B, Hashim M, & Khan N. Role of plant growth promoting rhizobacteria (PGPR) as a plant growth enhancer for sustainable agriculture: A review. Bacteria. 2024; 3(2): 59-75.
CrossRef - Saeed Q, Xiukang W, Haider FU, Kučerik J, Mumtaz MZ, Holatko J, Mustafa A. Rhizosphere bacteria in plant growth promotion, biocontrol, and bioremediation of contaminated sites: a comprehensive review of effects and mechanisms. International journal of molecular sciences. 2021; 22(19): 10529.
CrossRef - Lovley DR, Coates JD, Blunt-Harris EL, Phillips EJ, Woodward JC. Humic substances as electron acceptors for microbial respiration. Nature. 1996; 382(6590): 445-448.
CrossRef - Semhi K, Chaudhuri S, Clauer N, Boeglin JL. Impact of termite activity on soil environment: A perspective from their soluble chemical components. International Journal of Environmental Science & Technology. 2008; 5: 431-444.
CrossRef - Fall S, Brauman A, Chotte JL. Comparative distribution of organic matter in particle and aggregate size fractions in the mounds of termites with different feeding habits in Senegal: Cubitermes niokoloensis and Macrotermes bellicosus. Applied Soil Ecology. 2001; 17(2): 131-140.
CrossRef - Adebajo SO, Akintokun PO, Ezaka E, Ojo AE, Olannye DU, Ayodeji OD. Use of termitarium soil as a viable source for biofertilizer and biocontrol. Bulletin of the National Research Centre. 2021; 45: 1-8.
CrossRef - Fall S, Nazaret S, Chotte JL, Brauman A. Bacterial density and community structure associated with aggregate size fractions of soil-feeding termite mounds. Microbial ecology. 2004; 48: 191-199.
CrossRef - Lugtenberg BJ, Malfanova N, Kamilova F, Berg G. Microbial control of plant root diseases. Molecular microbial ecology of the rhizosphere. 2013; 1: 575-586.
CrossRef - Abdelhameed RE, Metwally RA, Soliman SA. Prospects of Bacillus amyloliquefaciens (MZ945930) mediated enhancement of Capsicum annuum plants under stress of Alternaria alternata in terms of physiological traits, thiol content, antioxidant defense, and phytohormones. Journal of Plant Growth Regulation. 2024 ; 43(9): 3265-3281.
CrossRef - Bibi G, Gagosh Nayyar B, Ajmal M, Mehak A, Seerat W, Shahbaz M, Akram A. Effect of culture filtrates of Alternaria alternata on seed germination and seedling growth of sesame. Archives of Phytopathology and Plant Protection. 2023; 56(8): 625-635.
CrossRef - Jayapradha C, Raja Y. A review of eco-friendly management of Alternaria species. Indian Journal of Hill Farming. 2016; 29(2).
- Kumar P, Dubey RC, & Maheshwari DK. Bacillus strains isolated from rhizosphere showed plant growth promoting and antagonistic activity against phytopathogens. Microbiological research. 2012; 167(8): 493-499.
CrossRef - Aneja KR. Experiments in microbiology, plant pathology, tissue culture and microbial biotechnology. New Age International Publishers. 2018; 5th Edition
- Schaeffer AB, Fulton MD. A simplified method of staining endospores. Science. 1933; 77(1990): 194-194.
CrossRef - Ismail MA, Amin MA, Eid AM, Hassan SED, Mahgoub HA, Lashin I, Fouda, A. Comparative study between exogenously applied plant growth hormones versus metabolites of microbial endophytes as plant growth-promoting for Phaseolus vulgaris Cells. 2021; 10(5): 1059.
CrossRef - Bakker A., & Schippers BOB. Microbial cyanide production in the rhizosphere in relation to potato yield reduction and Pseudomonas spp-mediated plant growth-stimulation. Soil Biology and Biochemistry. 1987; 19(4): 451-457.
CrossRef - Petatán-Sagahón I, Anducho-Reyes, MA, Silva-Rojas HV, Arana-Cuenca A, Tellez-Jurado A, Cárdenas-Álvarez IO, Mercado-Flores Y. Isolation of bacteria with antifungal activity against the phytopathogenic fungi Stenocarpella maydis and Stenocarpella macrospora. International Journal of Molecular Sciences. 2011; 12(9): 5522-5537.
CrossRef - Skidmore AM, Dickinson CH. Colony interactions and hyphal interference between Septoria nodorum and phylloplane fungi. Transactions of the British Mycological Society. 1976; 66(1): 57-64.
CrossRef - Tagg J, McGiven A. Assay system for bacteriocins. Applied microbiology. 1971; 21(5): 943-943.
CrossRef - Bric JM, Bostock RM, Silverstone SE. Rapid in situ assay for indoleacetic acid production by bacteria immobilized on a nitrocellulose membrane. Applied and environmental Microbiology. 1991; 57(2): 535-538.
CrossRef - Gordon SA, Weber RP. Colorimetric estimation of indoleacetic acid. Plant physiology. 1951; 26(1): 192.
CrossRef - Ismail MA, Amin MA, Eid AM, Hassan SED, Mahgoub HA, Lashin I, Fouda, A. Comparative study between exogenously applied plant growth hormones versus metabolites of microbial endophytes as plant growth-promoting for Phaseolus vulgaris Cells. 2021; 10(5): 1059.
CrossRef - Janardan Yadav JY, Verma JP, Tiwari KN. Effect of plant growth promoting rhizobacteria on seed germination and plant growth chickpea (Cicer arietinum) under in vitro conditions. Biological Forum. 2010; 2 (2): 15-18, 25
- López-Hernández D. Nutrient dynamics (C, N and P) in termite mounds of Nasutitermes ephratae from savannas of the Orinoco Llanos (Venezuela). Soil Biology and Biochemistry. 2001; 33(6): 747-753.
CrossRef - Jouquet P, Mamou L, Lepage M, Velde B. Effect of termites on clay minerals in tropical soils: fungus‐growing termites as weathering agents. European Journal of Soil Science. 2002; 53(4): 521-528.
CrossRef - Wood TG, Johnson RA, Anderson JM. Modification of soils in Nigerian savanna by soil-feeding Cubitermes (Isoptera, Termitidae). Soil Biology and Biochemistry. 1983; 15(5): 575-579.
CrossRef - Menaut JC, Barbault R, Lavelle P, Lepage M. African savannas: biological systems of humification and mineralization. Ecology and management of the world’s savannas. 1985; 14-33, 90
- Abbadie L, Lepage M. The role of subterranean fungus comb chambers (Isoptera, Macrotermitinae) in soil nitrogen cycling in a preforest savanna (Cote dIvoire). Soil Biology and Biochemistry. 1989; 21(8): 1067-1071.
CrossRef - Nikhil K. Termiticulture: environmental technology for new millennium. Asian journal of microbiology biotechnology and environmental sciences. 2006; 8(1): 93.
- Lavelle P, Spain AV. Soil organisms. Soil ecology. 2001; 201-356.
CrossRef - Jouquet P, Ranjard L, Lepage M, Lata JC. Incidence of fungus-growing termites (Isoptera, Macrotermitinae) on the structure of soil microbial communities. Soil Biology and Biochemistry. 2005; 37(10): 1852-1859.
CrossRef - Fall S, Hamelin J, Ndiaye F, Assigbetse K, Aragno, M, Chotte JL, Brauman A. Differences between bacterial communities in the gut of a soil-feeding termite (Cubitermes niokoloensis) and its mounds. Applied and Environmental Microbiology. 2007; 73(16): 5199-5208.
CrossRef - Manjula A, Sathyavathi S, Pushpanathan M, Gunasekaran P, Rajendhran J. Microbial diversity in termite nest. Current Science. 2014; 1430-1434.
- Miyagawa S, Koyama Y, Kokubo M, Matsushita Y, Adachi Y, Sivilay S, Oba S. Indigenous utilization of termite mounds and their sustainability in a rice growing village of the central plain of Laos. Journal of Ethnobiology and Ethnomedicine. 2011; 7: 1-6.
CrossRef - Akhtar K P, Saleem MY, Asghar M, Haq MA. New report of Alternaria alternata causing leaf blight of tomato in Pakistan. Plant pathology. 2004; 53(6): 816-816.
CrossRef - Soleimani MJ, Kirk W. Enhance resistence to Alternaria alternata causing potato brown leaf spot disease by using some plant defense inducers. Journal of plant protection research. 2012; 52(1).
CrossRef - Pasche JS, Wharam CM, Gudmestad NC. Shift in sensitivity of Alternaria solani in response to QoI fungicides. Plant disease. 2004; 88(2): 181-187.
CrossRef - Campo Arana RO, Zambolim L, Costa LC. Potato early blight epidemics and comparison of methods to determine its initial symptoms in a potato field. Revista Facultad Nacional de Agronomía Medellin. 2007; 60(2): 3877-3890.
- Horsfield A, Wicks T, Davies K, Wilson D, Paton S. Effect of fungicide use strategies on the control of early blight (Alternaria solani) and potato yield. Australasian Plant Pathology. 2010; 39: 368-375.
CrossRef - Chauhan AK, Maheshwari DK, Kim K, Bajpai VK. Termitarium-inhabiting Bacillus endophyticus TSH42 and Bacillus cereus TSH77 colonizing Curcuma longa: isolation, characterization, and evaluation of their biocontrol and plant-growth-promoting activities. Canadian journal of microbiology. 2016; 62(10): 880-892.
CrossRef - Heydari A, Pessarakli M. A review on biological control of fungal plant pathogens using microbial antagonists. Journal of biological sciences. 2010; 10(4): 273-290.
CrossRef - Ahmad F, Ahmad I, Khan MS. Screening of free-living rhizospheric bacteria for their multiple plant growth promoting activities. Microbiological research. 2008; 163(2): 173-181.
CrossRef - Omar AM, Ahmed AI. Antagonistic and inhibitory effect of some plant rhizo-bacteria against different Fusarium isolates on Salvia officinalis. American-Eurasian Journal of Agricultural and Environmental Sciences. 2014; 14(12): 1437-1446.
- Choudhary DK, Johri BN. Interactions of Bacillus spp. and plants–with special reference to induced systemic resistance (ISR). Microbiological research. 2009; 164(5): 493-513.
CrossRef - Panda AK, Bisht SS, DeMondal S, Senthil Kumar N, Gurusubramanian G, Panigrahi AK. Brevibacillus as a biological tool: a short review. Antonie Van Leeuwenhoek. 2014; 105: 623-639.
CrossRef - Hu HQ, Li XS, He H. Characterization of an antimicrobial material from a newly isolated Bacillus amyloliquefaciens from mangrove for biocontrol of Capsicum bacterial wilt. Biological control. 2010; 54(3): 359-365.
CrossRef - Ongena M, Jacques P. Bacillus lipopeptides: versatile weapons for plant disease biocontrol. Trends in microbiology. 2008; 16(3): 115-125.
CrossRef - Mukherjee AK, Das K. Correlation between diverse cyclic lipopeptides production and regulation of growth and substrate utilization by Bacillus subtilis strains in a particular habitat. FEMS microbiology ecology. 2005; 54(3): 479-489.
CrossRef - Otto M. Bacterial evasion of antimicrobial peptides by biofilm formation. Antimicrobial peptides and human disease. 2006; 251-258.
CrossRef - Bais HP, Fall R, Vivanco JM. Biocontrol of Bacillus subtilis against infection of Arabidopsis roots by Pseudomonas syringae is facilitated by biofilm formation and surfactin production. Plant physiology. 2004; 134(1): 307-319.
CrossRef - Barakat I, Zelmat L, Bidima, M G, Chtaina, N. In vitro and in vivo biological control of Alternaria alternata fungus by Bacillus spp. in Citrus fruit. Moroccan Journal of Agricultural Sciences. 2024; 5(3): 163-168.
- Choub V, Yim EY, Choi SI, Won SJ, Moon JH, Yun JY, Ahn YS. Effective biocontrol efficacy of Bacillus subtilis CV21 against cherry leaf spot disease caused by Alternaria phytopathogens and growth promotion of flowering cherry (Prunus sargentii Rehder) seedlings. Biological Control. 2024; 197: 105603.
CrossRef - Abbo AS, Idris MO, Hammad AM. The antifungal effects of four tomato rhizosphere Bacillus spp. against Alternaria alternata. International Journal of Science and Research. 2014; 3(7): 1324-8.
- Cazorla FM, Mercado-Blanco J. Biological control of tree and woody plant diseases: an impossible task?. BioControl. 2016; 61(3): 233-242.
CrossRef - Coley-Smith JR, Cooke RC. Survival and germination of fungal sclerotia. Annual review of phytopathology. 1971; 9(1): 65-92.
CrossRef - Agarwal M, Dheeman S, Dubey RC, Kumar P, Maheshwari DK, Bajpai VK. Differential antagonistic responses of Bacillus pumilus MSUA3 against Rhizoctonia solani and Fusarium oxysporum causing fungal diseases in Fagopyrum esculentum Microbiological research. 2017; 205: 40-47.
CrossRef - Bhattacharjee R, Dey U. An overview of fungal and bacterial biopesticides to control plant pathogens/diseases. African Journal of Microbiology Research. 2014; 8(17): 1749-1762.
CrossRef - Kloepper JW. Plant growth-promoting rhizobacteria on radishes. In Proc. of the 4th Internet. Conf. on Plant Pathogenic Bacter, Station de Pathologie Vegetale et Phytobacteriologie, INRA, Angers, France. 1978 ; 2: 879-882.
- Glick BR. The enhancement of plant growth by free-living bacteria. Canadian journal of microbiology. 1995; 41(2): 109-117.
CrossRef - Marques A P, Pires C, Moreira, H, Rangel AO, Castro PM. Assessment of the plant growth promotion abilities of six bacterial isolates using Zea mays as indicator plant. Soil Biology and Biochemistry. 2010; 42(8): 1229-1235.
CrossRef - Şahin F, Çakmakçi R, Kantar F. Sugar beet and barley yields in relation to inoculation with N2-fixing and phosphate solubilizing bacteria. Plant and soil. 2004; 265(1): 123-129.
CrossRef - Jeon JS, Lee SS, Kim HY, Ahn TS, Song HG. Plant growth promotion in soil by some inoculated microorganisms. The Journal of Microbiology. 2003; 41(4): 271-276
- Lucy M, Reed E, Glick BR. Applications of free living plant growth-promoting rhizobacteria. Antonie van leeuwenhoek. 2004; 86: 1-25.
CrossRef - Dey RKKP, et al. Growth promotion and yield enhancement of peanut (Arachis hypogaea L.) by application of plant growth-promoting rhizobacteria. Microbiological research. 2004; 159.4: 371-394.
CrossRef - Kannojia P, Choudhary KK, Srivastava AK, Singh AK. PGPR bioelicitors: induced systemic resistance (ISR) and proteomic perspective on biocontrol. In PGPR amelioration in sustainable agriculture. 2019 ; 67-84.
CrossRef - Lynch JM. Origin, nature and biological activity of aliphatic substances and growth hormones found in soil. In Soil organic matter and biological activity (Dordrecht: Springer Netherlands. 1985; 151-174.
CrossRef - Sagar A, Rai S, Sharma S, Perveen K, Bukhari NA, Sayyed RZ, Mastinu A. Molecular Characterization Reveals Biodiversity and Biopotential of Rhizobacterial Isolates of Bacillus Spp. Microbial Ecology. 2024; 87(1): 83.
CrossRef - Panchami PS, Geetha Thanuja K, Karthikeyan S. Isolation and characterization of indigenous plant growth-promoting rhizobacteria (PGPR) from cardamom rhizosphere. Current Microbiology. 2020; 77(10): 2963-2981.
CrossRef - Li Y, Zhang X, He K, Song X, Yu J, Guo Z, Xu M. Isolation and Identification of Bacillus subtilis LY-1 and Its Antifungal and Growth-Promoting Effects. 2023; 12(24): 4158.
CrossRef - Aziz ZFA, Saud HM, Rahim KA, Ahmed OH. Variable responses on early development of shallot (Allium ascalonicum) and mustard (Brassica juncea) plants to Bacillus cereus Malaysian Journal of Microbiology. 2012; 8(1): 47-50.
- Majeed A, Abbasi MK, Hameed S, Imran A, Rahim N. Isolation and characterization of plant growth-promoting rhizobacteria from wheat rhizosphere and their effect on plant growth promotion. Frontiers in microbiology. 2015; 6: 198.
CrossRef - Vashist H, Sharma D, Gupta A. A review on commonly used biochemical tests for bacteria. Innovare J Life Sci. 2013;1:1-7.
- Shoaib M, Muzammil I, Hammad M, Bhutta Z, Yaseen I. A mini-review on commonly used biochemical tests for identification of bacteria. Int J Res Publ. 2020;54.
CrossRef

