Introduction
Plants require a diverse array of important nutrient elements for the fulfillment of their life cycle. The 17 vital nutrients required are categorized into macro and micro-nutrients based on their concentrations in plant tissue, essential for promoting robust plant growth.1 The macro-nutrients form structural components of molecules and are required up to10 mmol/kg dry weight. The nine macro-elements required are Carbon (C), Hydrogen (H), Oxygen(O), Nitrogen(N), Potassium (K), Magnesium (Mg), Calcium (Ca), Sulfur (S) and Phosphorus (P). Some macro elements like Ca and Mg serve regulatory roles in addition to their structural role.1 Certain micronutrients, though required in minute amount (less than 10 mmol/kg of dry weight) are vital for supporting growth, development, and metabolic processes of plants.2 These micronutrients required in minute quantity perform crucial catalytic and regulatory functions, often acting as enzyme activators and include Chlorine (Cl), Boron (B), Iron (Fe), Manganese (Mn), Zinc (Zn), Copper (Cu), Nickel (Ni), and Molybdenum (Mo) as depicted in Fig 1. Sodium, Cobalt, Silicon, Selenium are the elements that play role in plant growth and development. These are known as Trace elements as they are present in very minute amount in plants. Despite being present in smaller quantities, micronutrients play a vital role in plant development by influencing the levels of phenolics and lignin, and also by contributing to the stability of the plasma membrane.3 Plants exist within a rich ecosystem teeming with diverse microbes in the rhizospheric region surrounding their roots. These microbes are incredibly important for plant health, going beyond simply helping with nutrient uptake. In natural ecosystems, most nutrients are present in the form of complex organic molecules or rocks which make them less bioavailable to plants. PGPRs show various enzymatic activities that break down and convert organic and inorganic nutrient, thereby increase their availability to plants. Besides, they also aid in breaking down recalcitrant nutrients into usable forms and producing organic acids and siderophores that increase the availability of specific nutrients. Many microbes also form symbiotic relationships with plants, like mycorrhizae, which extend their hyphae far beyond the root area, significantly enhancing the plant’s ability to absorb nutrients and water. Mycorrhizal fungi also enhance nutrient transport into plant cells by stimulating the genes associated with nutrient uptake. Similarly, nitrogen-fixing bacteria, provide a readily available source of nitrogen to the plant by forming symbiotic relationships. Analyzing root released—compounds by plant roots can help us to understand the complex communication between plants and microbes. The enormous potential of these microbial communities to replace current fertilization practices could be realized once their specific mechanisms are fully understood.
Factors such as soil pH, climate change, and lack of farmer awareness contribute to poor micronutrient availability, particularly of iron, zinc, and boron. This not only lowers crop yield and quality but also results in micronutrient-deficient food, contributing to “hidden hunger” in humans, particularly in developing regions that rely heavily on plant-based diets. Iron and zinc deficiencies are linked to anemia, impaired immunity, and stunted growth. Addressing these issues require sustainable strategies like improved soil management, biofortification, microbial interventions, and supportive policies to enhance crop nutrient density and protect global food and nutrition security.4 The strategic utilization of microbial strains through intellectual applications can lead to the production of nutrient-rich grains, addressing global under-nourishment and benefiting populations in need. The biofortification of nutrients in crops through effective PGPM is a novel, eco-friendly technique in addressing the problem of malnutrition. The rhizospheric niche showing an enormous diversity of microbial taxa5 as revealed through sequencing studies has become an exploring area in the plant-microbe interactions for beneficial purposes. There is a need for further research focused on exploring and documenting the diversity and complexity of the root microbiome through metagenomic approaches. With changing global climatic conditions, interest has also shifted more towards designing synthetic communities. These communities consist of strains representing the predominant rhizospheric taxa, aiming to replicate beneficial microbial functions by harnessing efficient microbes in controlled experimental settings.6 This review focuses on the microbes assisting in micro-nutrient mobilization and uptake by plants to sustain healthy growth.
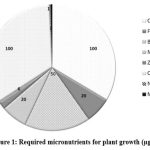 |
Figure 1: Required micronutrients for plant growth (µg/g).7 |
Rhizospheric niche
The rhizosphere constitutes an exceptionally intricate environment housing billions of microbes, predominantly bacteria and fungi. These microorganisms play a pivotal role in enhancing plant growth, with a particular emphasis on maintaining root health, thereby ensuring a sufficient supply of nutrients for overall plant development. Robust plants, in return, activate the soil microbial community by releasing root exudates and leaving behind organic residues. A plant’s root modifies the rhizospheric environment by exuding large amounts of carbon. The carbon exudated by the root leads to a huge increase in the rhizospheric microorganisms living both inside and outside the root system. These root exudates consist of complex mixtures of low-molecular-weight organic compounds such as organic acids, amino acids, sugars, and phenolics along with enzymes for the rhizosphere organisms. In some plants through roots, 30 to 50% of the photosynthetically assimilated carbon goes to the soil.8 The rhizospheric zone serves as a focal point for intricate biological interactions between plants and microbes. The wide range of interactions can be competitive, mutualistic, exploitative, commensal, or neutral. Previous studies on plant-microbe interactions have demonstrated the crucial role of rhizospheric microbes in alleviating the impacts of pathogens, herbivory9,10 and various abiotic stresses.11,12 Further research is crucial to unveil the mechanisms of micro-nutrient uptake, given their vital role in influencing plant growth. The three major ways by which the microbes influence plant growth are depicted in Fig 2.
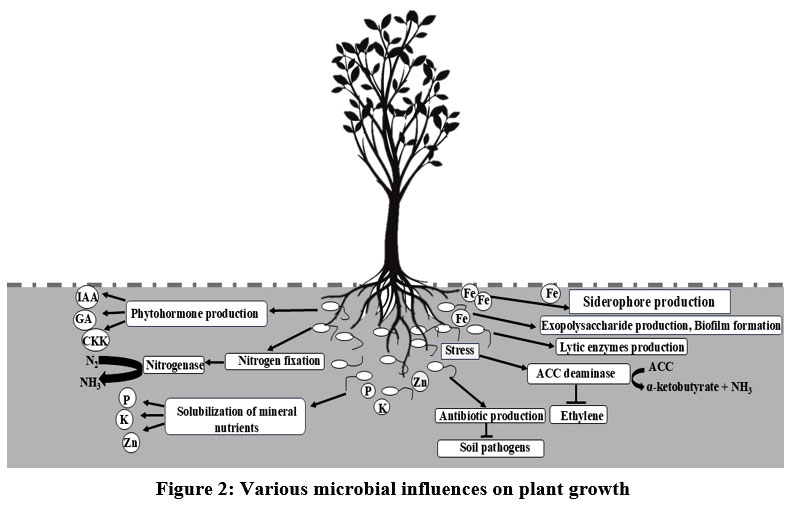 |
Figure 2: Various microbial influences on plant growth |
Mobilization of nutrients by microbes
Ecosystem productivity is mainly governed by the nutrient transformation ability of the microbes present in the soil.13 Metabolic processes within soil microbes enable the depolymerization and mineralization of nutrients bound in organic molecules, resulting in their release into the soil. The microbial cell contents are subsequently exudated into the soil through processes such as turnover, protozoic predation, or cell lysis.14,15 Increasing crop productivity by the addition of chemical fertilizers and fungicides for meeting the goals of food security has adversely affected the microbial flora, which is the key governing component in ecosystem productivity and stability. Addition of biostimulators or inhibitors along with traditional practices like tillage, manuring, residue placement, irrigation has influence on vital microbial processes. Chemical fertilizers and fungicides can be substituted by the growth-promoting rhizospheric microbes which get associated with roots of plants and stimulate plant productivity by uptake of nutrients in a soluble form. Microbial-mediated soil transformation takes place through various mechanisms like extracellular enzyme activity, acidic dissolution, altering the oxidation state of ions, siderophore production, chelation, mineralization, etc., which make nutrients available to plants (Table 1).
Table 1: Microbes associated in uptake of micronutrient and their role plant growth.
|
S. No |
Nutrient |
Uptake form |
Plant requirements |
Deficiency symptoms |
Soil system deficient |
Microbe in nutrient uptake and solubilization |
|
1 |
Manganese |
Mn2+ |
Photosynthesis, N-metabolism |
Interveinal chlorosis, brown necrotic spots, premature leaf drop, delay anther maturity |
Organic soil, high pH soil, sandy soil low organic matter, over-limed soils |
Glomus diaphanum, G. aggregatum, Bacillus megaterium, Penicillium citrinum |
|
2 |
Molybdenum |
MoO4-2 |
N & S-metabolism, protein synthesis, pollen formation |
Irregular leaf blade, interveinal mottling, marginal chlorosis |
Acid, sandy soils in humid regions |
Pseudomonas, Bacillus leptothrix, Citrobacter, Acetobacter, Nitrospira fermicutes |
|
3 |
Zinc |
Zn 2+ |
Protein synthesis, growth regulation |
Short internodes, decreased leaf size |
Sandy soil, organic soil |
Klebsiella spp, Serratia marcescense, Azotobacter, Azospirillum, Burkholderia Enterobacter, Anabaena, Calothrix, P. fluorescens, P. pudida, Bacillus aryabhattai, Pseudomonas |
|
4 |
Chloride |
Cl– |
Stomatal opening, photosynthetic reactions |
Wilting, branched root |
sandy, inland soil |
Bacillus megaterium, Hafnia alvei, Pantoea agglomerans. |
|
5 |
Boron |
Bo3-3 |
Cell wall formation, sugar transport, flowering, pollen formation and germination |
Rosetting, barren ears, hollow stem and fruit, discolored leaves, loss of fruiting bodies |
Acidic sandy soil in regions of high rain fall |
G. intraradices, Arthrobacter, Rhodococcus, Gracibacillu, Lysinibacillus, Algoriphagus, Enterococcus |
|
6 |
Copper |
Cu+, Cu2+ |
Carbohydrate and nitrogen metabolism, lignin synthesis, Vitamin A synthesis |
Stem and twig dieback, leaf yellowing, stunted development, and pale green foliage. |
Sandy soil low in organic matter |
G. mosseae, Burkholderia sp. GL12, Bacillus megaterium JL35 |
|
7 |
Iron |
Fe2+ |
Production of chlorophyll, lignin formation, nitrogen reduction and fixation |
Interveinal chlorolysis, Necrosis |
Sandy soil, high pH soil |
P. fluorescens, P aeruginosa, Thiobacillus ferrooxidans, Bacillus megaterium |
|
8 |
Nickel |
Ni2+ |
N-metabolism, seed germination, |
Hindered embryo growth, decreased leaf size, leaf tip necrosis |
High pH soils |
Sphingomonas macrogoltabidus, Microbacterium liquefaciens, Microbacterium arabinogalactanolyticum, |
Microbes release extracellular enzymes and organic compounds with the specific purpose of solubilizing plant-available nutrients from sources such as soil organic matter, crop and animal residues, or manures. Soil microbes produce organic acid and extracellular enzymes which help in solubilizing the nutrients fixed in organic material and soil minerals thereby speeding up the nutrient cycle. Microbes can enhance the solubility of nutrients like iron by deriving energy from oxidation and reduction reactions. The increased nutrient content in plants after application of PGPR is primarily attributed to their influence on the growth and development of roots. This, in turn, leads to enhanced nutrient uptake by the plants.16 Much work has been done to understand the role of microbes in N2 fixation and P-solubilization compared to other minerals. Certain specialized symbiotic bacteria like Rhizobium and Bradyrhizobium can fix atmospheric N2 into ammonium-based compounds for plant nutrition. There are also symbiotic N2-fixing bacteria like Frankia that associate with woody shrubs. Azospirillum, an asymbiotic bacterium, provides additional N by getting inoculated with roots of grasses of economic importance like sugarcane. N2 fixation is estimated to provide 10 to 20% of the N requirement in the case of cultivated crops and accounts for approximately 25 to 40% of the global annual demand for reactive nitrogen. The significance of mycorrhizal fungi and nodulated legumes were well realized long back from the second half of 19th century.17 By the 1950s, efforts to improve crop yield by coating seeds with PGPR cultures like Azotobacter chroococcum or Bacillus megaterium known as seed priming came into practice.18 By the 1980s many different rhizobacteria strains, with promising growth-promoting activities like Pseudomonas, and Azospirillum, were described as plant growth-promoting bacteria and since then many bacterial strains have been added to the list like Klebsiella aerogenes, Serratia marcescens, Enterobacter cloaceae.19
PGPRs have been extensively researched for their role in promoting plant growth through multiple mechanisms, including phytohormone production, nitrogen fixation, improvement of soil structure, decomposition of organic matter, recycling of vital nutrients, and solubilization of minerals such as phosphorus, potassium, and zinc. These activities significantly enhance crop growth, yield, and seed size, while also supporting soil fertility and overall soil health.20,21 PGPR produces high molecular weight carbohydrates known as exopolysaccharides which help in maintaining soil moisture under drought conditions.22 Inoculation of seedlings with high exopolysaccharide producing bacteria shows enhanced soil aggregation capability and the roots of such plants adhere to more soil and show higher relative water content in their leaves.23 Application of PGPR increases the antioxidant enzyme level in plants under severe water stress conditions and hence can be used as inoculants to reduce the oxidative damage elicited by water stress.24 Plants that have been inoculated with PGPR possessing ACC (1-aminocyclopropane-1-carboyclic acid) deaminase activity exhibits a comparatively higher tolerance to environmental stress.25 ACC serves as the precursor in ethylene production in plants. Bacteria that produce ACC deaminase can reduce ethylene levels by breaking down ACC into α-ketoglutarate and ammonia. This enzymatic action promotes root growth, as it lowers endogenous ethylene levels.26 The application of PGPR in the form of eco-friendly fertilizer has become a sustainable agricultural approach, providing an effective substitute for harmful chemical fertilizers. The application of mineral fertilizers, containing concentrated soluble nutrients affects the adjacent microbial niche. An osmotic stress on adjacent microbial environment is generated due to the presence of salt in higher concentration around a fertilizer granule until the diffusion of nutrients into the soil. Likewise, high pH around the microbial niche due to anhydrous ammonia temporarily hinders its activity. In contrast, the use of PGPR biofertilizers mitigates these concerns and promotes sustainable agricultural practices. A better perception of the contribution of PGPM in micronutrient uptake is required. This approach can aid in the effective management of microbial inoculums by pairing them with compatible host crops under optimized soil conditions. This includes ensuring sufficient soluble phosphorus (P) and molybdenum (Mo) levels, as well as adjusting soil pH, to enhance the overall efficacy of microbes in various cropping systems. The rhizospheric soil harbor essential soil enzymes and microorganisms which coordinate in the breakdown of organic matter, mineralization and nutrient cycling thereby enhancing nutrient availability for plant uptake. Addition of at least 10% of rhizospheric soil was found to promote vegetation reconstruction.27 The biosynthesis pathways of phytohormones generated or regulated by PGPMs have been better understood through developments in genomics, proteomics, and metabolomics. This has also highlighted the potentials of PGPMs in controlling vital physiological processes in plants. As microbes also secrete various toxins, antibiotics and enzymes which influence plant biochemistry, physiology, and secondary metabolisms, their application is a new and eco-friendly approach to control the proliferation of plant pathogens.28,29
Boron
Boron (B) is an essential micronutrient necessary for the growth and development of organisms ranging from diatoms to vascular plants. However, fungi, bacteria, green algae, and animals do not require B.30 Boron deficiency is a common global agricultural problem as 11.26 % of it in earth is in the form of borax. Plant uptake of B occurs passively when B availability is high, while under low B supply, the uptake becomes facilitated (with boric acid) or active (with borate). Boron deficiency in plants affects some of the critical physiological processes such as cell wall synthesis, sugar transport, lignin deposition, cell wall structure,31 metabolism of carbohydrates, RNA integrity, respiration, indole acetic acid secretion, phenol secretion, and membrane integrity. Recent studies have revealed that deficiency in vitamin B can potentially disrupt the metabolism of ascorbate metabolism32 as well. Adequate B in plants has been shown to minimize the chances of attack on crop plants by pathogenic fungi like Plasmodiophora brassicae infecting crucifers, Fusarium solani infecting beans, Verticillium albo-atrum infecting tomato and cotton, and many viruses like tomato yellow leaf curl virus in tomato, tobacco mosaic virus in bean, Gaeumannomyces graminis (Sacc.)33 and Blumeria graminis in wheat. Boron acquisition occurs easily in low pH soils as compared to high pH soils as the mannitol–B complex is stable in alkaline environment and checks its entry into plant cells. Few species of mycorrhiza like Glomus intraradices, G. etunicatum, and G. diaphanum irrespective of soil pH conditions, when inoculated in maize plants have been documented to enhance B transport to shoots and enhance the yield two folds as compared to uninoculated maize crops.34 Inoculation of Glomus fasciculatum in lemon (Citrus jambhiri Lush) plants grown in soil amended with B enhanced B accumulation from 11-18% in leaves, simultaneously enhancing the secretion of amino acids and root sugars, as compared to control plants.35 Bacillus boroniphylus a novel PGPR found in boron-rich soil promotes plant growth by producing organic acid which helps in solubilizing boron. B. boroniphylus is also recorded to protect plants from various stress factors.36 Boron is tightly held in organic matter and can be released into the soil solution after the breakdown of organic matter facilitated by Gypsophila, Bacillus, and Microbacter which sequester B by forming unreactive precipitate mineral form with Fe, Ca, or PO4, at pH 8-9. A few bacteria like Rhodococcus, Lysinibacillus, Gracibacillus, Arthrobacter, and Algoriphagus are much being worked out as efficient B solubilizers.37
Iron
Iron (Fe) is an important micro-nutrient that functions as a cofactor for numerous enzymes involved in key physiological processes like photosynthesis, respiration, and nitrogen fixation; therefore, its deficiency leads to significant metabolic disruptions. Iron primarily exists in its oxidized form (Fe³⁺), which leads to the formation of oxides and hydroxides that are insoluble, making them unavailable to plants and soil microorganisms. Plants can avail iron by two basic methods. The first method involves the chelation of iron through the release of organic compounds. This increases iron solubility further allowing it to diffuse toward the plant. Subsequently, it undergoes reduction and is absorbed by the plant through a membrane-associated enzymatic system. The second method involves the absorption of a reduced form of iron inside the plant by forming the complex formed between Fe+3 and organic compound.38 Some PGPRs are capable of releasing iron-chelating molecules called siderophore which attracts iron towards the rhizospheric part and makes them easily available for plant.39 Siderophores, the low-weight organic compounds of nearly less than 1kDa containing functional groups like hydroximates and catechols reversibly bind to iron. Estimated siderophore content in soil is around 10−30M. PGPRs such Pseudomonas aeruginosa and Pseudomonas fluorescens secretes pyoverdine and pyochelin type of siderophores.40 With more studies being carried out in this area the siderophore-producing bacteria enlisted has increased. Rhizobacteria Aeromonas, Arthrobacter, Bacillus, and Streptomyces produce Fe-chelating compounds or siderophores like hydroxate, catechol, and pyoverdins, which sequester Fe from soils. The stability of siderophores is highly pH dependent, (for example hydroxamate siderophores are stable at pH 3-4, while catechol is stable at pH 7.5-10).41 PGPRs can increase their competitive potential, in the rhizosphere as these compounds also have an antimicrobial activity besides improving iron nutrition in plants.42 Bacillus megaterium strain, showed 60-80% manganese and iron mobilization by its bio-solubilization capacity that were checked on polluted soil, whereas zinc was mobilized only by 20%.43 Results reveal that Azospirillum bacteria were capable of significantly increasing the Cu, Fe, Mn, and Zn content in maize leaves.44 Co-inoculation of Enterobacter, Serratia marcescens, and Microbacterium arborescens increased the micronutrient contents, including copper (Cu), zinc (Zn), manganese (Mn), and iron (Fe), in wheat grain under both pot and field experiments as compared to control.45
Manganese
In plants, manganese (Mn) functions as a cofactor, playing a role in the activation of around 35 different enzymes. The majority of these enzymes are involved in catalyzing decarboxylation, oxidation-reduction and hydrolytic reactions. Mn plays a structural role in enzymes, including the Mn-protein of Photosystem II and the Mn-dependent superoxide dismutase.46 As the oxidized forms of manganese are insoluble, plants can absorb it only in its reduced form, such as Mn²⁺. The oxidation-reduction state of manganese is influenced by soil pH. Soil having pH less than 6 reduction state exists and more than 6.5 Mn exists in its oxidation state. It was found that yield of wheat, oats, barley, sorghum, rice, and legumes were equally affected by both deficient and excessive levels of soil Mn. Manganese is the most well-known micronutrient for its ability of enhancing plant resistance to various root and foliar diseases such as blast of rice, wilt of tomato, common scab of potato, downy mildew, powdery mildew and all disease of wheat.47,48 Mn plays an important role in photosynthesis, secondary metabolite synthesis like lignin, phenolic compounds, and some other physiological mechanisms in plants49 that develops resistance against diseases.
Rhizospheric microbes by their redox activity considerably affect the Mn availability in soil. Some studies have shown that under Mn deficiency P. fluorescens and B. subtilis when inoculated in crops have been reported for increasing Mn uptake in crops which results in improving lignin production and defense mechanism of crops against some phytopathogens.50 Some more studies reveal that Bacillus cereus improves the yield parameters of wheat like seed size, biomass, and nutrient value of seed by enhancing the availability of Mn.51 Certain actinomycetes, along with species from the Streptomyces, Pseudomonas, and Bacillus genera, have also been found to enhance manganese availability in crops.52 Glomus diaphanum, G. aggregatum, Bacillus megaterium,53 and Penicillium citrinum54 play a significant role in enhancing manganese uptake by plants through their nutrient-solubilizing abilities.
Zinc
Zinc (Zn) is a vital micronutrient necessary for plant growth and reproduction. Insufficient levels of Zn can compromise membrane integrity, impede the production of carbohydrates, cytochromes, auxin, nucleotides, and chlorophyll, and make plants more susceptible to heat stress.55 It is also responsible for suppressing male fertility in plants.56 The available fraction of Zn in plants is very limited in soil due to its low persistency and high reactivity. In India, nearly 50% of soils are zinc-deficient, even though zinc is relatively abundant in the soils of the country’s semi-arid tropical regions.55
Application of inorganic zinc is not an effective solution as up-to 96–99% of applied Zn within 7 days of application becomes insoluble due to soil types and physicochemical reactions57 and thus becomes unavailable to plants. Excessive utilization of zinc containing fertilizers impairs the absorption of iron and copper. Zinc solubility is strongly influenced by soil pH and moisture levels, which makes the semi-arid areas often zinc-deficient. Soil pH plays a crucial role in determining the microelements availability and even a minor alteration in pH has a considerable effect, as lowering of pH influences Zn availability significantly in plants.58 Therefore, lowering the soil pH can enhance the availability of zinc to plants. Microbes facilitate the solubilization of metal forms through protons, oxidoreductive systems, and chelated ligands located on the surface of their cells. Organic acids released by rhizobacteria contain some protons which decreases the pH of rhizospheric soil59,60 that facilitates the mobility of Zn. PGPR produces gluconate or some gluconic acid derivatives like 2-keto gluconic acid as reported for Pseudomonas fluorescence,60 5-keto gluconic acids,61 and various organic acids62 which solubilize insoluble zinc. Bacillus sp. AZ6 demonstrates the ability to solubilize insoluble forms of zinc by secreting organic acids such as caffeic acid, ferulic acid, cinnamic acid, gallic acid, chlorogenic acid, and syringic acid in a liquid medium.63
Rhizobacteria comprising Rhizobium, Acinobacter, Pseudomonas, Bacillus, etc. are also considered proficient Zn-solubilizers. Bacillus spp. solubilizes ZnO, ZnS and ZnCO357 by altering the broth pH. Mostly wheat, rice, maize, oats, and cotton are affected by Zn deficiency.64 Many genera of both bacteria and fungi are known to improve zinc availability for plants and increase zinc content in cereal crops.65
The bioavailability of Zn in the rhizosphere has also been reported to increase by microbes secreting Zn-chelating compounds.66 Chelation has been demonstrated as the primary phenomenon to improve Zn availability in plant roots by Microbacterium saperdae, Pseudomonas monteilii and Enterobacter cancerogenesis.67 Pseudomonas Sp. (K-1, 96-51), Azospirillum lipoferum (JCM-1270, ER-20) and Azospirillum sp. (Ca-18) have been documented to solubilize fixed zinc from soil and makes it available for rice for longer duration by secreting chelating substance like ethylene diamine tetra acetic acid (EDTA).62 It has also been reported that Penicillium bilaji enhance Zn availability to plants through a chelation mechanism.68 PGPR such as Pseudomonas fluorescens, Klebsiella spp., and Serratia marchesense found in soil, solubilizes insoluble form of Zn to soluble form Pseudomonas.69,70 Treatment of seeds with some zinc solubilizing PGPR like Azospirillum, Pseudomonas, Burkholderia, Azotobacter and Acinetobacter strains have shown potential as plant growth promoters by enhancing attributes such as root volume, shoot height, leaf area, and dry biomass.71 Some of the effective Zn-enhancing bacterial strains reported in maize are Bacillus species AZ672 Pseudomonas,73 Azotobacter and Azospirillum.44 Bacillus aryabhattai strains MDSR7, MDSR11, and MDSR 14 have been shown to substantially mobilize Zn in soybean and wheat.74 A few cyanobacteria like Anabaena sp cw1, Providecie sp pw5, Anabaena sp. Cw3 and Calothrix sp. Cw2 has been reported to improve the nutrient quality of wheat grain by improving micronutrients like Zn, Fe, Cu and Mn0.75 Joshi et al.76 have reported a positive relationship between microbial inoculations and grain yield. The use of microbial inoculants offers multiple advantages, including increased zinc content in the flag leaf and a 50% reduction in nitrogen fertilizer requirements, which helps lower cultivation costs. The study indicated that comparable yields of wheat and maize can be achieved even with a 50% reduction in nitrogen fertilizer application.
Arbuscular mycorrhizal fungi are also found to lower the pH of rhizospheric soil, which contributes to the release of zinc from mineral fraction.77,65 Mycorrhizal fungi capable of solubilizing zinc prevent various fungal and bacterial diseases64 by elevating the Zn level in plants. According to the findings of Imran et al.,68 supplementing essential micronutrients—particularly zinc (Zn)—effectively mitigated growth inhibition and oxidative leaf damage in maize seedlings exposed to low root zone temperatures.78 Inoculation with PGPM, such as cyanobacteria, bacterial strains, and biofilms, can significantly enhance the concentration of micronutrients in the rhizosphere of different maize hybrids.79
Zn application in soil minimizes the chances of infections caused by Fusarium graminearum and Gaeumannomyces graminis in wheat. Zn protects plants by detoxifying the superoxide radicals that cause oxidative damage to membranes.80 Plants growing in calcareous and sodic soils contains large amount of insoluble ZnCO3 (zinc carbonate) that reduces Zn availability for plants. So, the plants growing in these regions suffer more with Zn deficiency as compared to others.81
Copper
Copper (Cu) is required in lignin synthesis as well as carbohydrate and nitrogen metabolism. It is also essential in photosynthesis and plant respiration. Due to its necessity for maintaining cell wall strength and preventing wilting; therefore, its deficiency in plants leads to symptoms like stunted growth, dieback of stems and twigs, and leaf yellowing, primarily due to impaired lignin production.
From a nutritional standpoint, mycorrhiza is the most prevalent and significant symbiotic association between roots and fungi residing in soil. Virtually all plants of economic importance form mycorrhizal association. This symbiotic relationship came into existence over 350 million years82 ago and has played beneficial roles in the colonization of land plants. Usually, the intra-radical mycelium of AM fungi profusely grows in the cortical region of the host plant roots. Extra radical arbuscular mycorrhiza hypha multiplies in the soil around the root and easily absorbs micro and macro nutrients such as Nitrogen, phosphorus, zinc, and copper and transports them into the host plant.83 Although well known for phosphorus uptake, the arbuscular mycorrhizal fungus also enhances the availability of other important micronutrients like Zn and Cu and macro element K.84 About 30 to 150% increase in dry weight is observed in tree seeding infected with AM. Enrichment of nutrients in plants is of concern where either the soil’s nutrient content is low or has heterogeneous distribution of nutrients.85 AM fungi are among the few significant soil microbes recognized for their ability to absorb nutrients with limited mobility (e.g. P, Zn, NH4+etc),86 as the extra radical AM hyphae can provide the nutrients by exploring additional soil regions extending beyond the nutrient-depleted zone formed around the root. Rhizobacteria that are active in the supply of Cu are Pseudomonas, Bacillus, Sphingomonas, Arthrobacter, Stenotrophomonas, etc, but are mostly studied in natural phytoremediation perspective in polluted soils.87 They help in maintaining homeostasis in the rhizosphere.
Molybdenum
In aqueous solution, Molybdenum (Mo) mainly occurs as molybdate ion MoO42-. Among all identified micronutrients, Mo has the lowest requirement in plants, primarily associated with its function in nitrogen metabolism. The enzymes like dinitrogenase and nitrate reductase require molybdenum for their function. A plant’s molybdenum requirement is therefore related to some degree to the form of nitrogen supply. The occurrence of molybdenum deficiency varies significantly among species, with a notable prevalence in legumes, Brassicaceae family members, and maize. This deficiency is intensified in acidic soils with elevated iron precipitates, as these soils strongly absorb the molybdate ion. Bacterial genera like Bacillus, pseudomonas, Citrobacter, Leptothrix, Acidobacter, Nitrospira and Firmicutes, are known to make Mo available to plants by a similar mechanism as Fe, by producing Mo-binding siderophores, and therefore, facilitating towards the uptake under limiting conditions.88 Limited information is accessible regarding molybdenum uptake in the cropping system.
Chlorine
Chloride ions (Cl–) are widely distributed and exhibit high solubility, making their deficiency a rare occasion. Manganese, in conjunction with chloride, is necessary for the oxygen-evolving process that takes place in photosynthesis. Chloride (Cl–) functions as an extremely mobile anion and serves as the primary counter-ion to diffusing cations, playing a crucial role in preserving electrochemical balance across cell membranes. It serves as the primary osmotically active solute within the vacuole and is essential for cell division. Research on the role of microbes in chloride uptake is limited as plants readily absorb chloride, and many accumulate chloride ions well beyond their minimal requirement. Chloride-deficient plants often show stunted growth, wilting at the tips of leaves, and widespread chlorosis.89 Bacillus megaterium and Pantoea agglomerans90 contribute to improved chloride uptake in plants by facilitating ion transport and enhancing root absorption efficiency.
Nickel
Nickel (Ni) is a plentiful metallic element that is readily absorbed by plant roots. Approximated 200ng of nickel is needed for a plant to go through its entire life cycle, a demand that is typically satisfied by the initial nickel content present in the seed.91 While the precise role of Ni in plants remains somewhat unclear, it is potentially linked to the nitrogen requirement during the germination of seeds. It is known to be a component of two enzymes—urease and hydrogenase. A deficiency in nickel can result in reduced urease activity in plants. Sphingomonas macrogoltabidus, Microbacterium liquefaciens, and Microbacterium arabinogalactanolyticum enhance nickel uptake in plants by promoting metal solubilization.92
Trace elements
Trace elements play an important role in plant nutrition. Despite being required by plants in minute quantities, elevated concentrations of these elements can be toxic to plants. Moreover, their lack and an excess both lead to severe disease symptom in plants. Rhizospheric microbes are mostly studied for land management in trace element contaminated soils. Trace elements like Selenium (Se), Sodium (Na), and Cobalt (Co) required by plants in trace amounts93 are discussed below.
Sodium
Sodium (Na) stimulates cell expansion leading to growth, and can partially replace potassium as an osmotically active solute. The accumulation of solutes such as K+ or Na+ inside the cell reduces its water potential and increases its solute potential which leads to enter the water into the cell. This results in an increase in turgor pressure. This pressure drives cell growth by promoting cell expansion, thereby contributing to growth at both the tissue and whole-plant levels.94 Mainly increase in sodium concentration is directly related to the salinity of soil.95 Saline stress in plants adversely impacts fundamental processes including photosynthesis, protein synthesis, as well as energy and lipid metabolism.96 White and Broadley97 reported that the quantity of salt in a cell more than it’s carrying capacity leads to cell dehydration and finally death.
Selenium
Selenium (Se) is a highly soluble and essential element for plants. At its low concentration, it shows free radicals scavenging activity by increasing antioxidant enzymes such as Superoxide Dismutase (SOD) activity. It contributes to the formation cell membrane and amending photosynthetic systems by improving the formation of chloroplast components. It also plays a role in mitigating abiotic stress factors such as cold,98 drought,99 temperature,100 and heavy metal101 in plants, although at high concentrations, it becomes toxic. It is reported that on application of Se (1 mg/L) pears and peaches, the quality of fruit was improved.102 Additionally, Se concentration, extending up to 5 mg/L, have been found beneficial for certain Se-accumulating species, including Spirulina platensis, red seaweed, and Pteris vittata.103 Thus, plants demonstrate a variation in its accumulation effects.
Cobalt
Cobalt (Co) is a component of various enzymes and proteins, plays an important role in plant metabolism. It is also an essential part of cobalamin (vitamin B12), which is necessary for the functioning of several enzymes involved in nitrogen (N2) fixation.104 Cobalt at its higher concentration interferes with the metabolism of plants. At its higher concentration, it inhibits seed germination, plant growth, and photosynthesis.105 Cobalt is considered both a heavy metal and a micronutrient, and its precise regulation is important for normal plant growth and development.
Conclusion
Optimizing nutrient availability is crucial for maximizing crop yield and quality. While traditional fertilization methods aim to achieve this, their effectiveness can be limited, and their production and application often lead to significant environmental concerns. Fortunately, a vast array of soil microbes offers a more environment friendly pathways to fulfill plant nutritional needs. Microbe-dependent pathways for micronutrient uptake in plants involve enhanced solubilization of insoluble micronutrients via organic acid secretion, siderophore production for iron chelation, enzymatic release of bound nutrients, and stimulation of root growth through phytohormones. Additionally, nitrogen-fixing microbes also contribute to nutrient supply through asymbiotic/symbiotic nitrogen fixation. In contrast, microbe-independent pathways in plants while solely being depend on plant roots for absorbing nutrients from the soil solution, is often limited by soil nutrient solubility, availability and plant vigour. These pathways include direct ion uptake, root exudation of organic acids, and root morphological adaptations to improve nutrient acquisition. It is notable that the rhizospheric microbes not only help in plant nutrient uptake, particularly the recalcitrant nutrients but also influence the soil properties. Analyzing the root exudates can help decrypt the communication between plants and microbes. Plant’s nutritional status actively influences its interactions with microbes, demonstrating the plant’s role in initiating or deterring these associations. By unravelling the intricate signaling networks among microbes within the root microbiome, we can gain a deeper understanding of how microbial enhancements to plant nutrition function, especially when plants face various biotic and abiotic stresses. This knowledge will be key to developing more sustainable and effective agricultural practices. Many breakthroughs have been made in identifying the beneficial microbes but the complex plant-microbe interaction needs to be addressed more particularly by comparative genomic approaches to identify plant genes that shape the rhizospheric microbiome. Major gaps lie in understanding these signaling and the metabolic activities they share, which needs to be addressed to fully utilize their potential.
Acknowledgement
The authors would like to acknowledge the necessary support provided under the IoE scheme of Banaras Hindu University.
Funding Sources
Financial support received by CSIR-HRDG under project File no.37WS (0051)/2023- 24/EMR-II/ASPIRE is duly acknowledged. Authors are also grateful to Banaras Hindu University for the support extended under the Institute of Eminence (IoE) scheme
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
All datasets generated or analyzed during this study are included in the manuscript.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Richa Raghuwanshi: Conceptualization, Funding Acquisition, Resources, Supervision
Sandeep Kumar Gupta: Writing – Original Draft.
Jay Kishor Prasad: Data Collection, Analysis,
Riddha Dey: Data Collection, Analysis, Writing – Review & Editing
Seema Devi: Data Analysis, Writing – Review & Editing
Surya Prakash Dube: Writing – Review & Editing
Yashoda Nandan Tripathi: Writing – Review & Editing
References
- Kumar S., Kumar S., Mohapatra T. Interaction between macro‐and micro-nutrients in plants. Front Plant Sci, 2021; 12:665583.
CrossRef - Saleem S., Mushtaq N.U., Rasool A., Shah W.H., Tahir I., & Rehman R. U. Plant nutrition and soil fertility: physiological and molecular avenues for crop improvement. In Sustainable plant nutrition. Academic Press, 2023; (pp. 23-49). ISBN 9780443186752.
CrossRef - Bhat B.A., Islam S.T., Ali A, Sheikh B.A., Tariq L, Islam S.U., & Hassan Dar T.U., 2020. Role of micronutrients in secondary metabolism of plants. Plant Micronutrients: Deficiency and Toxicity Management. Springer, Cham, 2020;311-29. ISBN 978-3-030-49856-6.
CrossRef - Dhaliwal S. S., Sharma V., & Verma G. Agronomic strategies for improving micronutrient use efficiency in crops for nutritional and food security. Input Use Efficiency for Food and Environmental Security. Springer, Singapore, 2021;123-156. ISBN 978-981-16-5199-1.
CrossRef - Bulgarelli D., Schlaeppi K., Spaepen S., Van Themaat E. V. L., & Schulze-Lefert, P. Structure and functions of the bacterial microbiota of plants. Annu Rev Plant Biol, 2013; 64: 807–838.
CrossRef - Busby P.E., Soman C., Wagner M.R., Friesen M.L., Kremer J., Bennett A., Morsy M., Eisen J.A., Leach J.E., & Dangl J.L. Research priorities for harnessing plant microbiomes in sustainable agriculture. PLOS Bio. 2017; 15(3):e2001793.
CrossRef - Salisbury F. B., & Ross C. W. Plant physiology. Wadsworth Publishing Co., Belmont, California, 1992; 682 pp. (ISBN 0-534-15162-0).
- Kuzyakov Y., & Domanski G. Carbon input by plants into the soil. Review. J Plant Nutr Soil Sci, 2000; 163(4):421-431.
CrossRef - Chenghua L., Yijun He., Yaping C. Rhizosphere microbiome regulation: Unlocking the potential for plant growth. Current Research in Microbial Sciences, 2025, 8: 100322.
CrossRef - Pan X, Yue Y, Zhao F, Song T, Xu B, Li Z, Qi Z, Yu J, Cao H, Yu M, Shen Q, Xu J, Xiong Wu, Liu Y. Rhizosphere microbes facilitate the break of chlamydospore dormancy and root colonization of rice false smut fungi. Cell Host and Microbe. 2025 33 (5), 731-744.
CrossRef - Yaish M.W., Al-Lawati A., Jana G.A., Vishwas Patankar H., & Glick B.R. Impact of soil salinity on the structure of the bacterial endophytic community identified from the roots of caliph medic (Medicago truncatula). PLOS ONE, 2016;11(7): e0159007.
CrossRef - Meena V.S., Meena S.K., Verma J.P., Kumar A., Aeron A., Mishra P.K., Bisht J.K., Pattanayak A., Naveed M., & Dotaniya M.L. Plant beneficial rhizospheric microorganism (PBRM) strategies to improve nutrients use efficiency. Ecol Eng, 2017; 8(3): 107.
CrossRef - Schimel J.P., & Bennett J. Nitrogen mineralization: Challenges of a changing paradigm. Ecol, 2004; 85(3): 591-602.
CrossRef - Bonkowski M. Protozoa and plant growth: the microbial loop in soil revisited. New Phytol, 2004; 162(3): 617–631.
CrossRef - Richardson A.E., Barea J.M., McNeill A.M., & Prigent-Combaret C. Acquisition of phosphorus and nitrogen in the rhizosphere and plant growth promotion by microorganisms. Plant Soil, 2009; 305–339.
CrossRef - Canbolat M.Y., Bilen S., Çakmakçı R., Şahin F., & Aydın A. Effect of plant growth promoting bacteria and soil compaction on barley seedling growth, nutrient uptake, soil properties and rhizosphere microflora. Biol Fertil Soils, 2006; 42:350–357.
CrossRef - Scheublin T.R., Van Der Heijden M.G. Arbuscular mycorrhizal fungi colonize non-fixing root nodules of several legume species. New Phytol, 2006;172(4):732-738.
CrossRef - Mahmood A., Turgay O.C., Farooq M., & Hayat R. Seed biopriming with plant growth promoting rhizobacteria: a review. FEMS Microbiol Ecol, 2016;92(8): fiw112.
CrossRef - Gupta S.K., Prasad J.K., & Raghuwanshi R. Characterizing rhizospheric plant growth promoting bacteria for their effects on oat (Avena sativa). Int J Pharma Bio Sci, 2017; 8(4): 142-151.
CrossRef - Kohler J., Caravaca F., Carrasco L., & Roldan A. Contribution of Pseudomonas mendocina and Glomus intraradices to aggregate stabilization and promotion of biological fertility in rhizosphere soil of lettuce plants under field conditions. Soil Use Manag, 2006; 22(3): 298–
CrossRef - Prasad J.K., Gupta S.K., & Raghuwanshi R. Screening multifunctional plant growth promoting rhizobacteria strain for enhancing seed germination in wheat (Triticum aestivum). Int J Agri Res, 2017; 12(2): 64-72.
CrossRef - Hafsa N., & Asghari B. Role of plant growth promoting rhizobacteria and their exopolysaccharide in drought tolerance of maize. J Plant Interact, 2014; 9(1): 689–701.
CrossRef - Sandhya V.Z.A.S., SK. Z A., Grover M., Reddy G. and Venkateswarlu B.S.S.S. Alleviation of drought stress effects in sunflower seedlings by the exopolysachrides producing Pseudomonas putida strain GAP-P45. Biol Fertil Soils, 2009; 46: 17-26.
CrossRef - Sinha S., & Raghuwanshi R. Synergistic effect of Arbuscular mycorrhizal fungi and mycorrhizal helper bacteria on physiological mechanism to tolerate drought in Eclipta prostrata (L.). J Pure Applied Microbiol, 2016; 10(2):1117-1129.
- Raghuwanshi R., & Prasad J.K. Perspectives of rhizobacteria with ACC Deaminase activity in plant growth under abiotic stress. In: Giri B., Prasad R., Varma, A. (eds) Root Biology. Soil Biology, Springer, Cham, 2018; 52: 303-321. ISBN 978-3-319-75910-4.
CrossRef - Glick Bernard R. “Modifying a plant’s response to stress by decreasing ethylene production.” Phytoremediation Rhizoremediation. Dordrecht: Springer Netherlands, 2006; 227-236.
CrossRef - Cao Y., Ai Z., Dang X., Chen Q., Wang T., Li J., Deng Y., Liu H., Li Q., & Zhao M. The addition of rhizosphere soil can affect the plant-soil-microbial nutrients of Bothriochloa ischaemum. Rhizosphere, 2025 :101042.
CrossRef - Espinosa-Palomeque B., Jiménez-Pérez O., Ramírez-Gottfried RI., Preciado-Rangel P., Buendía-García A., Sifuentes GZ., Sariñana-Navarrete MA., & Rivas-García T. Biocontrol of Phytopathogens Using Plant Growth Promoting Rhizobacteria: Bibliometric Analysis and Systematic Review. Horticulturae, 2025; 11(3): 271.
CrossRef - Wu X., Liu Y., Jia B., Tao L., Li H., Wang J., Yuan Z., Sun X., & Yao Y. Four Decades of Bacillus Biofertilizers: Advances and Future Prospects in Agriculture. Microorganisms, 2025; 13(1): 87.
CrossRef - Lehto T., Ruuhola T., & Dell B. Boron in forest trees and forest ecosystems. For Ecol Manag, 2010; 260(12):2053-69.
CrossRef - O’Neill M.A., Ishii T., Albersheim P., & Darvill A.G. Rhamnogalacturonan II: Structure and function of a borate cross-linked cell wall pectic polysaccharide. Annu Rev Plant Biol, 2004; 55: 109–139.
CrossRef - Han S., Chen L.S., Jiang H.X., Smith B.R., Yang L.T., & Xie C.Y. Boron deficiency decreases growth and photosynthesis, and increases starch and hexoses in leaves of citrus seedlings. J plant physiol, 2008;165(13):1331-41.
CrossRef - Ojha R.K., & Jha S.K. Role of mineral nutrition in management of plant diseases. In: Singh HK, Solankey SS, Roy MK (eds) Farmers’ Prosperity through Improved Agricultural Technologies. Jaya Publishing House, New Delhi, 2021; pp 241–261
- Clark R.B., & Zeto S.K. Iron accession by mycorrhizal maize grown on alkaline soil. J Plant Nutr, 1996; 19(2):247-264.
CrossRef - Dixon R.K., Garrett H.E., & Cox G.S. Boron fertilization vesicular arbuscular mycorrhizal colonization and growth of citrus s jombhiri lush. J Plant Nutr, 1989;12(6): 687-700.
CrossRef - Ahmed I., Yokota A., & Fujiwara T. A novel highly boron tolerant bacterium, Bacillus boroniphilus nov., isolated from soil, that requires boron for its growth. Extremophiles, 2007; 11: 217–224.
CrossRef - Sen S., Mondal N., Ghosh W., & Chakraborty R. Inducible boron resistance via active efflux in Lysinibacillus and Enterococcus isolates from boron-contaminated agricultural soil. Biometals, 2022;1-14.
CrossRef - Colombo C., Palumbo G., He J.Z., Pinton R., & Cesco S. Review on iron availability in soil: interaction of Fe minerals, plants, and microbes. J Soil Sediment, 2014; 14:538-48.
CrossRef - Arora NK., Tewari S., & Singh R. Multifaceted plant-associated microbes and their mechanisms diminish the concept of direct and indirect PGPRs. In Plant microbe symbiosis: Fundamentals and advances. New Delhi: Springer India, 2013; 411-449.
CrossRef - Haas, & DéfagoG.Biological control of soil-borne pathogens by fluorescent pseudomonads. Nat Rev Microbiol, 2005;3(4): 307–319.
CrossRef - Chincholkar S.B., Chaudhari B.L., Talegaonkar S.K., & Kothari, R.M. Microbial iron chelators: a tool for sustainable agriculture. In: Upadhayay RK, Mukherji KG, Chamola BP (eds) Biocontrol potential and their exploration in crop disease management, Vol I. Kluwer Academic, New York, 2000; 49–70. ISBN 978-1-4613-6889-2.
CrossRef - Jha C.K., & Saraf M. Plant growth promoting rhizobacteria (PGPR). J Agric Res Dev, 2015;5(2):108-19. https://doi.org/10.1007/s00344-016-9663-5
CrossRef - Stefanescu I.A., Mocanu R., & Duncianu M. Bio-solubilization capacity of Bacillus Megatrium strain of some micronutrients from polluted soil. Cercetari agronomic in moldova, 2010; 43(2):142.
CrossRef - Biari A., Gholami A., & Rahmani H.A. Growth promotion and enhanced nutrient uptake of maize (Zea mays ) by application of plant growth promoting rhizobacteria in arid region of Iran. J Biol Sci, 2008;8(6):1015-1020
CrossRef - Kumar A., Maurya B.R., Raghuwanshi R., Meena V.S., & Tofazzal Islam M. Co-inoculation with Enterobacter and rhizobacteria on yield and nutrient uptake by wheat (Triticum aestivum) in the alluvial soil under Indo-Gangetic Plain of India. J Plant Growth Regul, 2017;36 (3): 608–617.
CrossRef - Tewari R.K., Kumar P., & Sharma P.N. Oxidative stress and antioxidant responses of mulberry (Morus alba) plants subjected to deficiency and excess of manganese. Acta Physiologiae Plantarum, 2013; 35:3345-56.
CrossRef - Heckman J.R., Sims J.T., Beegle D.B., Coale F.J., Herbert S.J., Bruulsema T.W. & Bamka W.J., Nutrient removal by corn grain harvest. Agron J, 2003;95: 587–591.
CrossRef - Simoglou K., & Dordas C. Effect of foliar applied boron, manganese and zinc on tan spot in winter durum wheat. Crop Prot, 2006; 25(7):657–663.
CrossRef - Farzadfar S., Zarinkamar F., & Hojati M. Magnesium and manganese affect photosynthesis, essential oil composition and phenolic compounds of Tanacetum Plant Physiol Biochem, 2017; 112:207-17.
CrossRef - De Schamphelaire L., Rabaey K., Boon N., Verstraete W., & Boeckx P. Minireview: The potential of enhanced manganese redox cycling for sediment oxidation. Geomicrobiol J, 2007; 24, 7(8): 547–558.
CrossRef - Ijaz A., Mumtaz M.Z., Wang X., Ahmad M., Saqib M., Maqbool H., Zaheer A., Wang, W., & Mustafa A. Insights into manganese solubilizing Bacillus for improving plant growth and manganese uptake in maize. Front Plant Sci, 2021; 12:719504.
CrossRef - Nogueira M.A., Nehls U., Hampp R., Poralla K., & Cardoso E.J.B.N. Mycorrhiza and soil bacteria influence extractable iron and manganese in soil uptake by soybean. Plant soil, 2007;298: 273-284.
CrossRef - Zhang W. H., He L. Y., Wang Q., & Sheng X. F. Inoculation with endophytic Bacillus megaterium 1Y31 increases Mn accumulation and induces the growth and energy metabolism-related differentially-expressed proteome in Mn hyperaccumulator hybrid Pennisetum. hazard. Mater, 2015; 300:513-521.
CrossRef - Acharya C., Kar R. N., & Sukla, L. B. Bioleaching of low-grade manganese ore with Penicillium citrinum. J. Miner. Process. Environ. Prot, 2002; 2(3):197-204.
- Singh B., Natesan S.K.A., Singh B.K., Usha K. Improving zinc efficiency of cereals under zinc deficiency. Curr Sci, 2005;88(1): 36–44.
CrossRef - Frassinetti S., Bronzetti G.L., Caltavuturo L., Cini M., & Della Croce, C. The role of zinc in life: a review. J Environ Pathol Toxicol Oncol, 2006; 25(3):597-603.
CrossRef - Saravanan V.S., Subramoniam S.R., & Raj S.A. Assessing in vitro solubilization potential of different zinc solubilizing bacterial (ZSB) isolates. Braz J Microbiol, 2004;35(1-2): 121–125.
CrossRef - Saleem M. H., Usman K., Rizwan M., Jabri H. A., Alsafran M. Functions and strategies for enhancing zinc availability in plants for sustainable agriculture. Plant Sci., 2022;13. https://doi.org/10.3389/fpls.2022.1033092
CrossRef - Wu S.C., Cheung K.C., Luo Y.M., & Wong M.H. Effects of inoculation of plant growth promoting rhizobacteria on metal uptake by Brassica Environ Pollut, 2006;140(1): 124–135.
CrossRef - Fasim F., Ahmed N., Parsons R. & Gadd G.M. Solubilization of zinc salts by bacterium isolated by the air environment of tannery. FEMS Microbiol Lett, 2002; 213(1):1–6.
CrossRef - Saravanan V., Madhaiyan M., & Thangaraju M. Solubilization of zinc compounds by the diazotrophic, plant growth promoting bacterium Gluconacetobacter diazotrophicus. Chemosphere, 2007;66(9):1794–1798.
CrossRef - Tariq M., Hameed S., Malik K.A., & Hafeez F.Y. Plant root associated bacteria for zinc mobilization in rice. Pak J Bot, 2007;39(1): 245–253.
- Hussain D., Haydon M.J., Wang Y., Wong E., Sherson S.M., Young J., Camakaris J., Harper J.F., & Cobbett, C.S. P-type ATPase heavy metal transporters with roles in essential zinc homeostasis in Arabidopsis. Plant Cell, 2004;16(5):1327–1339.
CrossRef - Rashid A., & Ryan J. Micronutrient constraints to crop production in the Near East: Potential significance and management strategies. In: Micronutrient Deficiencies in Global Crop Production. Springer, Dordrech, 2008;149–180.
CrossRef - Subramanian K.S., Tenshia V., Jayalakshmi K., & Ramachandran V. Biochemical changes and zinc fractions in arbuscular mycorrhizal fungus (Glomus intraradices) inoculated and uninoculated soils under differential. Appl Soil Ecol, 2009; 43(1):32-39.
CrossRef - Obrador A., Novillo J., & Alvarez J.M. Mobility and availability to plants of two zinc sources applied to a calcareous soil. Soil Sci Soc Am J, 2003; 67(2):564–572.
CrossRef - Whiting S.N., Souza M.D., & Terry N. Rhizosphere bacteria mobilize Zn for hyper accumulation by Thlaspi caerulescens. Environ Sci Technol, 2001;35(15):3144–3150.
CrossRef - Imran M., Arshad M., Khalid A., Kanwal S., & Crowley D.E. Perspectives of rhizosphere microflora for improving Zn bioavailability and acquisition by higher plants. Int J Agric Biol, 2014;16(3):653–662.
CrossRef - Upadhyay A., & Srivastava S. Characterization of a new isolate of Pseudomonas fluorescens strain Psd as a potential biocontrol agent. Lett Appl Microbiol, 2008;47(2): 98-105.
CrossRef - Upadhyay A., & Srivastava S. Evaluation of multiple plant growth promoting traits of an isolate of Pseudomonas fluorescens strain Psd. Indian J Exp Biol, 2010; 48: 601-609.
- Vaid S.K., Kumar B., Sharma A., Shukla A.K., & Srivastava P.C. Effect of Zn solubilizing bacteria on growth promotion and Zn nutrition of rice. J Soil Sci Plant Nutr, 2014;14(4): 889–910.
CrossRef - Hussain A., Arshad M., Zahir Z.A., & Asghar, M. Prospects of zinc solubilizing bacteria for enhancing growth of maize. Pak J Agric Sci, 2015; 52(4): 915–
- Goteti P.K., Emmanuel L.D.A., Desai S., & Shaik M.H.A. Prospective zinc solubilising bacteria for enhanced nutrient uptake and growth promotion in Maize (Zea mays ). Int J Microbiol, 2013;2013(1):869697.
CrossRef - Ramesh A., Sharma S.K., Sharma M.P., Yadav N., & Joshi, O.P. Inoculation of zinc solubilizing Bacillus aryabhattai strains for improved growth, mobilization and biofortification of zinc in soybean and wheat cultivated in vertisols of central India. Appl Soil Ecol, 2014;73: 87-96.
CrossRef - Rana A., Joshi M., Prasanna R., Shivay Y.S. & Nain L. Biofortification of wheat through inoculation of plant growth promoting rhizobacteria and cyanobacteria. Eur J Soil Biol, 2012;50: 118-126.
CrossRef - Joshi D., Negi G., Vaid S. & Sharma A. Enhancement of wheat growth and Zn content in grains by zinc solubilizing bacteria. IJAEB, 2013;6(3):363-370.
CrossRef - Martino E., Perotto S., Parsons R., & Gadd G.M. Solubilization of insoluble inorganic zinc compounds by ericoid mycorrhizal fungi derived from heavy metal polluted sites. Soil Biol Biochem, 2003;35(1):133–141.
CrossRef - Ghanbari Zarmehri S., Moosavi S.G., Zabihi H.R., & Seghateslami M.J. The effect of plant growth promoting rhizobacteria (PGPR) and zinc fertilizer on forage yield of maize under water deficit stress conditions. J Appl Sci Eng Technol Educ, 2013;3(23): 3281-3290.
- Prasanna R., Rana A., Chaudhary V., Joshi M., and Nain L. Cyanobacteria-PGPR Interactions for Effective Nutrient and Pest Management Strategies in Agriculture. In: Satyanarayana, T., Johri, B. (eds) Microorganisms in Sustainable Agriculture and Biotechnology. Springer, Dordrecht, 2012;173-195.
CrossRef - Cakmak I., Tansley Review No. 111 Possible roles of zinc in protecting plant cells from damage by reactive oxygen species. 2000; 146(2):185–205.
CrossRef - Hafeez B., Khanif M.Y., Saleem M. Role of zinc in plant nutrition. Am J Exp Agric, 2013;3(2): 374-391.
CrossRef - Van Der Heijden M.G., Martin F.M., Selosse M.A., & Sanders I.R. Mycorrhizal ecology and evolution: the past, the present, and the future. New phytol, 2015;205(4):1406-23.
CrossRef - Smith SE, Read D Mycorrhizal symbiosis, 3rd edn. London: Academic Press. 2010.
- Cavagnaro T.R., Jackson L.E., Six J., Ferris H., Goyal S., Asami D., & Scow K.M. Arbuscular mycorrhizas, microbial communities, nutrient availability, and soil aggregates in organic tomato production. Plant Soil, 2006; 282: 209-25.
CrossRef - Cavagnaro T.R., Smith F.A., Smith S.E., & Jakobsen I. Functional diversity in arbuscular mycorrhizas: exploitation of soil patches with different phosphate enrichment differs among fungal species. Plant Cell Environ, 2005;28(5):642-650.
CrossRef - Tinker P.B., Nye PH. Solute movement in the rhizosphere. Oxford University Press, Oxford, Great Britain, 2000.
CrossRef - Khan W.U., Ahmad S.R., Yasin N.A., Ali A., & Ahmad A. Effect of Pseudomonas fluorescens RB4 and Bacillus subtilis 189 on the phytoremediation potential of Catharanthus roseus (L.) in Cu and Pb-contaminated soils. Int J Phytoremediation, 2017;19(6): 514–521.
CrossRef - Gonzalez Ibarra A.A., Yanez Barrientos E., Wrobel K., Corrales Escobosa A.R. & Wrobel K. Effect of copper and molybdenum in nutrient solution on Cu, Mo, Fe, Mg, Ca, Zn, Na, K status in sunflower. J Plant Nutr, 2023;46(5):714-30.
CrossRef - Mondal S., & Bose B. Impact of micronutrient seed priming on germination, growth, development, nutritional status and yield aspects of plants. J Plant Nutr, 2019;42(19):2577-99.
CrossRef - Gond S. K., Torres M. S., Bergen M. S., Helsel Z., & White Jr J. F. Induction of salt tolerance and up-regulation of aquaporin genes in tropical corn by rhizobacterium Pantoea agglomerans. Lett Appl Microbiol, 2015;60(4):392-399.
CrossRef - Rahman H., Sabreen S., Alam S., & Kawai S. Effects of nickel on growth and composition of metal micronutrients in barley plants grown in nutrient solution. J Plant Nutr, 2005;28(3):393-404.
CrossRef - Abou‐Shanab R. A., Angle J. S., Delorme T. A., Chaney R. L., Van Berkum P., Moawad H., Ghanem K., & Ghozlan H. A. Rhizobacterial effects on nickel extraction from soil and uptake by Alyssum murale. New phytol, 2003; 158(1): 219-224
CrossRef - Vatansever R., Ozyigit II., & Filiz E. Essential and beneficial trace elements in plants, and their transport in roots: a review. Appl Biochem Biotechnol, 2017; 181:464-82.
CrossRef - Nieves-Cordones M., Al Shiblawi FR., & Sentenac H. Roles and transport of sodium and potassium in plants. The alkali metal ions: Their role for life. 2016:291-324.
CrossRef - Kumari A., Patel M., & Parida A. K. Physiological, Proteomics, and Metabolomics Approaches for Elucidation of Salinity and Alkalinity Tolerance in Halophytes. In Physiology of Halophytes. Apple Academic Press. 2025; 289-335.
CrossRef - Parida A.K., & Das A.B. Salt tolerance and salinity effects on plants: a review. Ecotoxicol Environ Saf, 2005;60(3):324-49.
CrossRef - White P.J., & Broadley M.R. Chloride in soils and its uptake and movement within the plant: a review. Ann Bot, 2001;88(6):967-88.
CrossRef - Chu J., Yao X., & Zhang Z. Responses of wheat seedlings to exogenous selenium supply under cold stress. Biol Trace Elem Res, 2010; 136:355-63.
CrossRef - Hasanuzzaman M., & Fujita M. Selenium pretreatment upregulates the antioxidant defense and methylglyoxal detoxification system and confers enhanced tolerance to drought stress in rapeseed seedlings. Biol Trace Elem Res, 2011; 143:1758-76.
CrossRef - Djanaguiraman M., Prasad P.V., & Seppanen M. Selenium protects sorghum leaves from oxidative damage under high temperature stress by enhancing antioxidant defense system. Plant Physiol and Biochem, 2010;48(12):999-1007.
CrossRef - Kumar M., Bijo A.J., Baghel R.S., Reddy C.R.K., & Jha, B. Selenium and spermine alleviate cadmium induced toxicity in the red seaweed Gracilaria dura by regulating antioxidants and DNA methylation. Plant Physiol Biochem, 2012; 51:129-38. https://doi.org/10.1016/j.plaphy.2011.10.016
CrossRef - Pezzarossa B., Remorini D., Gentile M.L., & Massai R. Effects of foliar and fruit addition of sodium selenate on selenium accumulation and fruit quality. J Sci Food Agric, 2012;92(4), 781–786.
CrossRef - Belokobylsky A.I., Ginturi E.I., Kuchava N.E., Kirkesali E.I., Mosulishvili L., Frontasyeva M.V., Pavlov S.S. & Aksenova N.G. Accumulation of selenium and chromium in the growth dynamics of Spirulina platensis. J Radioanal Nucl Chem, 2004; 259:65-8.
CrossRef - Hu X., Wei X., Ling J. & Chen J. Cobalt: an essential micronutrient for plant growth? Front Plant Sci, 2021; 12:768523.
CrossRef - Wei W., Wang Y., Wei Z.G., Zhao H.Y., Li H.X. & Hu F. Roles of organic acids and nitrate in the long-distance transport of cobalt in xylem saps of Alyssum murale and Trifolium subterraneum. Biol Trace Elem Res, 2009; 131:165-76.
CrossRef
Abbreviations List
|
PGPR |
Plant Growth-Promoting Rhizobacteria |
|
PGPM |
Plant Growth-Promoting Microorganisms |
|
ACC |
(1-aminocyclopropane-1-carboyclic acid) |
|
ROS |
Reactive Oxygen Species |
|
SOD |
Superoxide Dismutase |

