Introduction
There is a growing concern about the increasing demands on agricultural production and its future needs.1 Maize is a widely grown plant globally, and its consumption is 61% for feed, 17 % for food, and 22% for industrie.2-4 Non-thermal plasma and its effects upon the pre-treatment of maize seeds before sowing helps in improving the overall yield of the crops, and is one of the important methods employed in agriculture. The Plasma pre-treatment of maize seed employs both chemical as well as physical methods. The non-invasive physical methods being employed for seed priming include pre-sowing treatment of seeds with low magnetic fields and non-thermal plasma.5,6 Exposure of seeds to non-thermal plasma has resulted in improved germination of seeds and early growth of seedlings in wheat, maize, rye, barley, and soybean.7-9 Promotion in germination and seedling overall growth after treatment with plasma is attributed to physical changes on the surface of the seeds, which enhance hydrophilicit.10 Exposure to nonthermal plasma with atmospheric air gives rise to reactive oxygen species and nitrogen species (ROS and RNS).11 In wheat seeds treated with plasma, the concentration of H2O2 is enhanced, and H2O2 acts as a signalling molecule and activates the hydrolases. Starch is the major reserve food material in maize seeds, and although ROS do not directly degrade starch, they can generate signalling molecules like H2O2 and NO that can modulate the activity of enzymes like α amylase responsible for the degradation of starch.12,5 In the present study, the biochemical changes produced by pre-treating seeds with the N2, He, H2, and Ar plasma have been investigated. The level of H2O2 and the activity of α-amylase have been monitored after optimizing the gaseous plasma exposure to obtain maximum enhancement of seedling growth.
Material and methods
Maize Seed material
Maize (Zea mays variety GSF-2) seeds were purchased from Jain Seed stores located in Indore, Madhya Pradesh, India. The maize seeds exhibiting uniformity in shape and size were surface sterilized using 1% Mercuric chloride for ten minutes and subsequently rinsed with deionised water. The maize seeds were placed on no.1 filter paper (Whatman) moistened using 10 ml of deionised water in glass Petri dishes (15 cm in diameter) and kept in darkness at 26°C for a period of 0 to 120 hours. Biochemical assessments were carried out at 24-hour intervals during this time frame.
Plasma treatment
Gaseous Plasma was produced using an RF plasma machine (Figure 1). A 30-liter evacuated stainless steel cylinder with a 30 cm height and 36 cm diameter formed the plasma chamber. A Rotary Pump, 13.56 MHz, and 600 W RF power supply, was connected to the chamber. Before gas (N2, He, Ar, and H2) was introduced inside the chamber to produce the plasma, a vacuum of 4 x 10-2 m bar was created inside the chamber. For each exposure, 100 dry seeds were placed on a metal gauze and placed inside the chamber for the required amount of time to be exposed to plasma.
After incubating the plasma-treated and untreated control seeds in darkness at 26°C for eight days, the growth of seedlings in terms of length and fresh weight was measured. The healthy seedlings with good and well-developed epicotyls and radicals were used for measuring the length and fresh weight. The method developed by Abdul-Baki et al. (1973)12 was used to calculate the vigor of the seedlings.
α-Amylase assay
The method of Sawhney et al. (1970)13 was used for measuring activity of α-amylase enzyme (EC 3.2.1.1) in germinating maize seedlings (100 mg) from the time they were soaked in water up to 96 hours. After peeling the seed tissue, 100 mg of seedlings was crushed in a chilled pestle mortar using 5 ml of ice-cold acetone, at 80 percent was used to make the homogenate of the seed tissue. Homogenate was then centrifuged for ten minutes at 10,000 rpm in cooling centrifuge, keeping the temperature at 4°C. After decanting the supernatant, pellet was mixed in 0.02 M sodium phosphate buffer and centrifuged for 20 minutes at 12000 rpm at 4°C. α-amylase activity was measured with the collected supernatant. Reaction mixture containing 200 µl enzyme extract and 0.02 M of sodium phosphate buffer (PH 6.4) and 0.1 Normality of HCl, 0.1 % Starch was incubated for ½ hour. The reaction was stopped by 0.1 N of HCl, and the colour developed by 0.1% of I2KI. Then absorbance was noted and measured at 660 nm in a Shimadzu UV-Vis spectrophotometer (Model: UV-1900).
Hydrogen Peroxide Assay
Hydrogen peroxide content was estimated following the method of Mukherjee et al. (1983).14 Fresh seed tissue (125 mg) was ground in a chilled mortar and pestle, and homogenate was extracted using 2.5 mL of 1% (w/v) trichloroacetic acid (TCA). The extract was centrifuged at 12,000 rpm for fifteen minutes at 4 °C, and the clear supernatant is collected.
The hydrogen peroxide–titanium complex was formed by mixing 2.5 mL of supernatant with 2 mL of titanium reagent. The titanium reagent is prepared by dissolving 1 g of TiO₂ in 10 g of K₂SO₄ by adding 2 mL of ammonium solution, which facilitates the formation of a reactive titanium salt. After mixing, solution was centrifuged at 10,000 rpm for fifteen minutes to pellet the titanium–peroxide complex. The resulting precipitate is dissolved in 2.5 mL of 2 M H₂SO₄. The intensity of the yellow colour developed was measured at 415 nm using a Shimadzu UV–Visible spectrophotometer (Model UV-1900). The hydrogen peroxide content was calculated and expressed in µmol H₂O₂ per gram fresh weight.
Protein assay
Maize seedlings (0.5–1.0 g) were ground to a fine powder. Proteins were extracted in 5–10 mL extraction buffer (50 mM sodium phosphate, pH 7.0; 100 mM NaCl; 1 mM CaCl₂; 1% PVP; 0.1% Triton X-100) per g tissue. Homogenates were centrifuged at 12,000 × g for fifteen minutes at 4 °C, and supernatant was used for further processing. Total soluble protein was estimated by the method given by Lowry et al. (1951)15 using bovine serum albumin as standard; absorbance was noted at 750 nm, and protein concentration was made from standard curve. All determinations were performed in triplicate. Activity for α-amylase was calculated as per mg protein.
Statistical analysis
Samples were drawn at random from ten seedlings receiving the same treatment for each trial.
The mean ± SEM of triplicate is used to express all of the information. All experiments are carried out with minimum of three replicates. All germination and biochemical parameters were conducted in 3 separate experiments with 3 replicated measurements. Data are presented as mean ± SEM and was examined using one-way analysis of variance. (ANOVA) following the post hoc Newman–Keuls multiple comparison test (*P<0.05, ** P<0.01, *** P<0.001).
Results
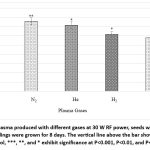 |
Figure 1: Effect of plasma produced with different gases at 30 W RF power, seeds were exposed for 240 seconds, and seedlings were grown for 8 days. The vertical line above the bar shows the SEM. |
Pre-sowing treatment of maize seeds with different gaseous plasmas, namely nitrogen, helium, hydrogen, and argon, for 240 seconds with 30 W power, indicated promotion in seedling length compared to untreated control seedlings after eight days of growth. The maximum promotion was 55.6% with N2, 47.4% with He, 26.8% with H2, and with Ar gas 18.5 % in comparison to untreated control seedlings (Figure 1).
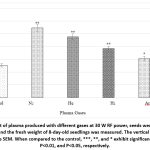 |
Figure 2: Effect of plasma produced with different gases at 30 W RF power, seeds were exposed for 240 seconds, and the fresh weight of 8-day-old seedlings was measured. The vertical line above the bar shows the SEM. |
Fresh weight of 8-day-old seedlings grown after plasma-treated seeds also showed promotion. In Comparison to untreated control seedlings, fresh weight was promoted by 113%(N2), 85%(He), 5O%(H2), and 21%(Ar). (Figure. 2).
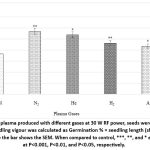 |
Figure 3: Effect of plasma produced with different gases at 30 W RF power, seeds were exposed for 240 seconds, and seedling vigour was calculated as Germination % × seedling length (shoot + root). The vertical line above the bar shows the SEM. |
Seedling Vigor, based on percent germination multiplied by seedling length (at 8DAE) showed an improvement with all the gases used (Figure.3) The promotion was higher in N2(56%) and He (47%) compared to H2 (29%) and Ar (19%) (Figure .3).
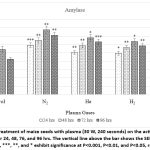 |
Figure 4: Effect of pretreatment of maize seeds with plasma (30 W, 240 seconds) on the activity of α-amylase after incubation of seeds for 24, 48, 76, and 96 hrs. The vertical line above the bar shows the SEM. |
Table 1: Percent increase in hydrolytic enzyme α-amylase after pre-treatment with gaseous plasma.
| Treatment gases | N2 | He | H2 | Ar |
| Incubation in hrs | ||||
| 24 | 62.8 | 38.38 | 34.36 | 21.51 |
| 48 | 54.71 | 40.5 | 36.76 | 24.86 |
| 72 | 65.43 | 42.69 | 37.28 | 30.14 |
| 96 | 43.87 | 32.18 | 27.44 | 20.33 |
Exposure of maize seeds to plasma enhanced the activity of α-amylase (Figure.4, Table 1). Enhancement was higher with N2 and He plasma and lower with H2 and Ar plasma at all stages (24 to 96 hours of incubation) compared with untreated control seedlings (Figure 4, Table). The peak in α-amylase activity was recorded after 72 hrs of incubation, both in the plasma-treated and untreated seedlings.
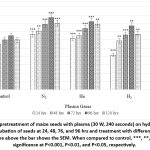 |
Figure 5: Effect of pretreatment of maize seeds with plasma (30 W, 240 seconds) on hydrogen peroxide in seedlings after incubation of seeds at 24, 48, 76, and 96 hrs and treatment with different gaseous plasma. The vertical line above the bar shows the SEM. |
The level of H2O2 was analysed in plasma-treated and untreated maize seeds after incubation of seeds from 24 to 120 hrs in darkness (Figure.5). The level of H2O2 gradually increased from 24 up to 96 hrs of incubation and then started decreasing at 120 hrs. The level of H2O2 was enhanced by exposure of seeds to plasma (Figure.5). N2 and H2 enhanced the level of H2O2 to a maximum of 83% and 66%, respectively, at 96 hrs (Figure.5, Table 2). H2 and Ar were less effective at 20% and 15% respectively at 96 hrs (Figure.5, Table 2)
Table 2: Percent increase in H2O2.
| Plasma Gases | N2 | He | H2 | Ar |
| 24 hrs | 24.71 | 19.68 | 10.96 | 2.34 |
| 48 hrs | 28.49 | 24.24 | 16.75 | 3.82 |
| 72 hrs | 43.35 | 33.15 | 18.14 | 8.25 |
| 96 hrs | 83.69 | 66.16 | 20.56 | 15.32 |
| 120 hrs | 38.38 | 30.47 | 11.56 | 8.71 |
The results demonstrated that pre-sowing plasma-treated seeds enhance subsequent growth of seedlings in the following order N2 > He>H2> Ar. The enhancement in the growth is preceded by a higher level of H2O2 and an enhanced activity of α-amylase from 24 to 72 hrs after the incubation of plasma-treated seedlings in darkness.
Discussion
Priming of seeds using plasma pre-treatment has earlier been done on wheat and maize and has been related to the sterilization of seed surfaces by exposure to plasma16-19 (Filatova,2011; Azad,2009; Miransari, 2014; Smith,2014). There were reports on higher absorption of water by plasma-treated seeds due to a partially etched seed surface, following exposure of seeds to plasma. There have been very few studies on the biochemical changes in the germinating seedlings after pre-treatment with non-thermal plasma. Results presented here indicate that treatment of seeds with plasma enhances the activity of the hydrolysing enzyme α-amylase that can hydrolyse starch stored in the seedlings. This degradation leads to higher seedling vigour of plasma-treated seeds. the enhancement in the α-amylase activity is preceded by an increase in the level of H2O2 produced in the germinating seeds. H2O2 acts as a signalling molecule, which may lead to the production of NO, as proved in magnetic field pre-treatment of maize seeds (Patel et al., 2017).5 Higher levels of H2O2 have been detected earlier in plasma-treated wheat seeds (Laroussi et al., 2004). Results presented indicate quantitative variation in the impact of plasma; the extent of promotion of growth of the seedlings depends upon the type of gas used in producing plasma. In the present study, N2 and He proved to be more effective than H2 and Ar. There is a correlation between the extent of promotion of the growth, the level of H2O2, and the enhancement of α-amylase activity with all the gases used to produce plasma.
Conclusions
The Pre-treatment of maize seeds with the non-thermal plasma is an effective method for enhancing seedling vigour. There are biochemical changes in the level of H2O2 and the activity of α-amylase in the plasma-treated seeds. There is a relationship between the growth of seedlings and the early biochemical changes recorded in the seedlings. In our present study, we presented that plasma pre-treatment significantly improves seedling vigour, and this enhancement is closely associated with early biochemical responses such as increased H₂O₂ levels and elevated α-amylase activity. These changes indicate that plasma exposure initiates a controlled oxidative signalling environment in the seed, which subsequently promotes germination and early growth. A key observation of this work is the superior effectiveness of N₂ and He plasmas compared with H₂ and Ar. This difference suggests that the biological impact of plasma is strongly influenced by the gaseous chemistry and the reactive species generated. N₂ plasma is known to produce a richer profile of reactive nitrogen species (RNS), including nitric oxide (NO), nitrite, and nitrate. NO is a well-established plant signalling molecule that interacts with reactive oxygen species (ROS) such as H₂O₂ to regulate germination, stress responses, and enzymatic activation. The synergy between plasma-generated H₂O₂ and RNS likely amplifies downstream signalling pathways, particularly those linked to α-amylase activation and starch mobilization. This mechanism provides a more robust explanation for the enhanced germination performance observed under N₂ plasma treatment. The strong effect of the plasma can be attributed to its high ionization potential, which facilitates the formation of long-lived metastable species. These metastable efficiently transfer energy to surrounding air molecules, generating higher levels of ROS and RNS compared to Ar plasma. This creates a more chemically reactive environment that supports favourable surface modifications, improved water uptake, and activation of early metabolic processes in the seedlings. In contrast, H₂ and Ar plasmas generate fewer biologically influential reactive species, which likely accounts for their comparatively weaker effects. By integrating the biochemical data with plasma chemistry, the present findings support a mechanistic model in which ROS–RNS interactions regulate early germination processes, including α-amylase induction and reserve mobilization. The quantitative relationship observed between these biochemical changes and seedling growth further reinforces the idea that plasma exerts its effects through signalling-driven physiological adjustments rather than direct stress or damage. Plasma pre-treatment emerges as a promising and efficient method for enhancing seedling vigour. The observed improvements are strongly linked to early biochemical changes—specifically, increased H₂O₂ accumulation and higher α-amylase activity—which collectively promote better germination and early growth. The superior performance of N₂ and He plasmas highlights the importance of gas-specific reactive species, particularly the role of RNS and NO (in the case of N₂) and enhanced energy-transfer-driven ROS/RNS formation (in the case of He). The key findings provide a clearer physiological basis for the differential effectiveness of plasma types. The study demonstrates that plasma-induced ROS–RNS signalling plays a central role in regulating seed metabolism and germination. Moving forward, future research should aim to characterize the exact ROS/RNS signatures generated by different gases, identify the downstream signalling pathways involved, and assess long-term impacts on plant development and stress tolerance. This work will help develop plasma treatment as a scalable, reliable, and tunable seed-priming technology for agriculture.
Acknowledgment
The authors truly thank every one of their coauthors for their invaluable effort. Their knowledge, commitment, and teamwork have been crucial to the accomplishment of this work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed consent statement
This study did not involve human participants, and therefore, informed consent was not required.
Author Contributions
Manisha Thakur: Writer, Resources, Conceptualization, Data Curation, Data analysis,Editing
Kadur Narayan Guruprasad: Supervision, Review
Uttam Sharma: Supervision
Kundan Viliya: Formal Analysis
References
- Abdul-Baki AA, Anderson JD. Vigor determination in soybean seed by multiple criteria. Crop Sci. 1973; 13:630–633. doi:10.2135/cropsci1973.0011183X001300060013
CrossRef - Azad MAK, Bae JH, Kim JS, Lim JK, Song KS, Shin BS, Kim HR. Isolation and characterization of a novel thermostable α-amylase from Korean pine seeds. New Biotechnol. 2009;26(3–4):143–149. doi: 10.1016/j.nbt.2009.09.006
CrossRef - Selcuk AM, Oksuz L, Basaran P. Decontamination of grains and legumes infected with Aspergillus spp. and Penicillium spp. by cold plasma treatment. Bioresour Technol. 2008; 99:5104–5109. doi: 10.1016/j.biortech.2007.09.076
CrossRef - Šerá B, Špatenka P, Šerý M, Vrchotová N, Hrušková I. Influence of plasma treatment on wheat and oat germination and early growth. IEEE Trans Plasma Sci. 2010; 38:2963–2968. doi:10.1109/TPS.2010.2060728
CrossRef - Nellemann C, MacDevette M, Manders T, et al. The Environmental Food Crisis and the Environment’s Role in Averting Future Food Crises. Birkeland Trykkeri AS; 2009.
- Filatova I, Azharonok V, Kadrov M, Beljavsky V, Govorov A, Shik A, Antonuk A. Effect of plasma treatment of seeds of some grain and legumes on their sowing quality and productivity. Rom J Phys. 2011.
- Di Girolamo G, Barbanti L. Treatment conditions and biochemical processes influencing seed priming effectiveness. Ital J Agron. 2012;7: e25. doi:10.4081/ija. 2012.e25
CrossRef - Galhaut L, Lespinay AD, Walker DJ, Bernal MP, Correal E, Lutts S. Seed priming of Trifolium repens L. improved germination and early seedling growth on heavy metal contaminated soil. Water Air Soil Pollut. 2014; 225:1905. doi:10.1007/s11270-014-1905-1
CrossRef - Laroussi M, Leipold F. Evaluation of the roles of reactive species, heat, and UV radiation in the inactivation of bacterial cells by air plasmas at atmospheric pressure. Int J Mass Spectrom. 2004; 233:81–86. doi: 10.1016/j.ijms.2003.11.016
CrossRef - Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951; 193:265–275.
CrossRef - Dhayal M, Lee SY, Park SU. Using low-pressure plasma for Carthamus tinctorius L. seed surface modification. Vacuum. 2006; 80:499–506.
CrossRef - Miransari M, Smith DL. Plant hormones and seed germination. Environ Exp Bot. 2014; 99:110–121. doi: 10.1016/j.envexpbot.2013.11.005
CrossRef - Mukherjee SP, Choudhuri MA. Implications of water stress-induced changes in the levels of endogenous ascorbic acid and hydrogen peroxide in Vigna seedlings. Physiol Plant. 1983;58(2):166–170. doi:10.1111/j.1399-3054. 1983.tb04162.x
CrossRef - El-Mougy NS, Abdel-Kader MM. Long-term activity of bio-priming seed treatment for biological control of faba bean root rot pathogens. Australas Plant Pathol. 2008; 37:464–471. doi:10.1071/AP08043
CrossRef - Patel P, Narayanaswamy GK, Kataria S, Baghel L. Involvement of nitric oxide in enhanced germination and seedling growth of magneto primed maize seeds. Plant Signal Behav. 2017;12(12): e1293217. doi:10.1080/15592324.2017.1293217
CrossRef - Sarath G, Hou G, Baird LM, Mitchell RB. Reactive oxygen species, ABA, and nitric oxide interactions on the germination of warm-season C4-grasses. Planta. 2007;226(3):697–708. doi:10.1007/s00425-007-0517-0
CrossRef - Sawhney S, Toky KL, Nanda KK. Changes in α-amylase activity during extension growth and floral induction in Impatiens balsamina, a qualitative short-day plant. Indian J Plant Physiol. 1970; 13:198–204.
- Zhou Z, Huang Y, Yang S, Chen W. Introduction of a new atmospheric pressure plasma device and application on tomato seeds. J Agric Sci. 2011; 2:23–30.
CrossRef

